- Home Page
- Company Profile
-
Our Products
- Active Pharmaceutical Ingredient
- (R)-Salbutamol Hydrochloride
- Veterinary APIs
- Pharmaceuticals impurities
- Pharmaceutical pellets
- 10 Hydroxycamptothecin
- 11-Alpha-Acetoxyprogesterone
- Aceglutamide
- 6-Dehydronandrolone Acetate
- Abacavir
- 3 Hydroxy 4 Amino Butyric Acid
- Acebrophylline
- 2,4-Dichlorobenzoic Acid
- 2',3'-Dideoxyuridine
- 2-Methoxyestradiol
- 3-Hydroxy-4-amino-butyric Acid
- 2-Nitroaminoimidazoline
- 3-O-Ethyl-L-ascorbic Acid
- 5-Androstenediol
- 5,7-Dichloro-8-hydroxyquinoline
- 5-Fluorouracil
- 5-hydroxy Tryptophan
- 5-Methyl-2'-deoxycytidine
- 8(4-Sulfophenyl)Theophylline
- 15S-Cloprostenol
- 1000px-Aspirin-skeletal.svg
- Abacavir Hydrochloride
- Abamectin
- Abarelix
- Abiraterone
- Abiraterone Acetate
- Acamprosate Calcium
- Acarbose
- Acebutolol
- Aceclofenac
- Acefylline Piperazine
- Acenocoumarol
- Acepifylline
- Acesulfame K
- Acetamide
- Acetazolamide
- Acexamic Acid
- Aciclovir Sodium
- Acipimox
- Acofide trihydrate
- Acotiamide
- Acriflavine hydrochloride
- Actinospectacin sulfate
- Adamantane
- Adapalene
- Adefovir
- Adefovir dipivoxil
- Ademetionine
- Ademetionine 1,4-butanedisulfonate
- Ademetionine disulfate tosylate
- Adenine
- Adenosine
- Adenosine triphosphate
- Adrenaline
- Afatinib Base
- Afatinib dimaleate
- Agomelatine
- Aica hydrochloride
- Alacepril
- Alanyl-glutamine
- Alarelin
- Albendazole
- Alendronate sodium trihydrate
- Alfuzosin
- Alfuzosin hydrochloride
- Algestone acetophenide
- Aliskiren
- Alizapride hydrochloride
- Almotriptan Maleate
- Alogliptin benzoate
- Alovudine
- Alpha-Asarone
- alpha-Ketovaline calcium
- Alpha Picolinic acid
- Altizide
- Altrenogest
- Amiloride hydrochloride
- Amitraz
- Amlexanox
- Amlodipine hydrochloride
- Ammonium bifluoride
- Ammonium fluoride
- Amodiaquine hydrochloride
- Amorolfine hydrochloride
- Amoxicillin Trihydrate
- Amphenidone
- Ampicillin
- Ampicillin sodium
- Ampicillin Trihydrate
- Amrinone
- Amygdalin
- Amyl Meta Cresol
- Anacetrapib
- Anastrozole Ingredient
- Anidulafungin
- Anisomycin
- Ansamitocin
- Antazoline Hydrochloride
- Antazoline Phosphate
- Antide
- Apomorphine Hydrochloride
- Apremilast
- Arformoterol Tartrate
- Argireline
- Aripiprazole
- Artemether.svg
- Artemisinin
- Articaine
- Articaine Hydrochloride
- Ascorbic Acid
- Ascorbyl Palmitate
- Asenapine
- Asenapine Maleate
- Aspartame
- Asunaprevir
- Atorvastatin Calcium
- Atorvastatin Calcium Trihydrate
- Atosiban
- Atosiban Acetate
- Atovaquone
- Atracurium
- Atracurium Besylate
- Atropine Sulfate
- Avatrombopag
- Avermectin
- Avibactam
- Avibactam Sodium
- Aviptadil
- Axitinib
- Azamethiphos
- Azasetron
- Azasetron hydrochloride
- Azathioprine
- Azathramycin
- Azelastine hydrochloride
- Azilsartan kamedoxomil
- Azilsartan medoxomil
- Azithromycin dihydrate
- Azlocillin sodium
- Azosemide
- Aztreonam
- Baccatine III
- Bacitracin Methylene Disalicylate
- Baicalin
- Balofloxacin
- Balsalazide disodium dihydrate
- Bambuterol hydrochloride
- Bamifylline Hydrochloride
- Bazedoxifene
- Bazedoxifene acetate
- Bedaquiline
- Bedaquiline fumarate
- Bendamustine
- Bendamustine hydrochloride
- Bendazol
- Bendroflumethiazide
- Benfotiamine
- Bengenin
- Benidipine hydrochloride
- Benproperine phosphate
- Benserazide hydrochloride
- Benzalkonium chloride
- Benzarone
- Benzbromarone
- Benzethonium Chloride
- Benzidamine hydrochloride
- Benziodarone
- Benzobarbital
- Benzocaine
- Benzonatate
- Benzphetamine Hydrochloride
- Benzthiazide
- Benztropine mesylate
- Benzylpenicilline Benzathine
- Benzyl Alcohol
- Benzylpenicillin Potassium
- Bepotastine Besilate
- Beraprost
- Beraprost Sodium
- Berberine hydrochloride
- Besifloxacin hydrochloride
- beta-Carotene
- Betahistine
- Betahistine dihydrochloride
- Betahistine Hydrochloride
- Betaine Citrate Anhydrous
- Betamethasone 17-valerate
- Betamethasone acetate
- Betamethasone Dipropionate
- Betamethasone Sodium Phosphate
- Betamethasone Valerate
- Betaxolol
- Hydroxycamptothecin
- Pilocarpine Hydrochloride
- Salbutamol hydrochloride
- Betaxolol hydrochloride
- Betulinic Acid
- Bevantolol Hydrochloride
- Bezafibrate
- Bicalutamide
- Bietamiverine
- Bifendatatum
- Bifendate
- Bifonazole
- Bilastine
- Bilirubin
- Biperiden Hydrochloride
- Biperiden Lactate
- Biphenyldicarboxylate
- Biphenyl Dimethyl Dicarboxylate
- Bisoprolol Fumarate
- Bisoprolol Fumerate
- Bisoprolol Hemifumarate
- Biuret
- Bivalirudin
- Bleomycin Hydrochloride
- Bleomycin Sulfate
- Blonanserin
- Bortezomib
- Bosentan
- Bosentan Monmonohydrateohydrate
- Bosutinib
- Brimonidine Tartrate
- Brinzolamide
- Brodimoprim
- Bromaminic Acid
- Bromfenac Monosodium Salt Sesquihydrate
- Bromfenac Sodium
- Bromhexine Hydrochloride
- Bromoacetonitrile
- Bromocriptine Mesylate
- Brompheniramine Maleate
- Buclizine
- Buclizine Dihydrochloride
- Budesonide
- Buflomedil Hydrochloride
- Bumetanide
- Bunitrolol
- Bupivacaine Base
- Buserelin
- Buserelin Acetate
- Buspirone
- Buspirone Hydrochloride
- Busulfan
- Butafosfan
- Butamirate citrate
- Butenafine hydrochloride
- Butizide
- Butoconazole
- Butopiprine
- Butorphanol Tartrate
- Butylamine
- Butylated hydroxyanisole
- Cabazitaxel
- Cabergoline
- Cabozantinib
- Cadazolid
- Caffeine
- Caffeine anhydrous
- Calamine
- Calcifediol
- Calcipotriol
- Calcipotriol anhydrous
- Calcipotriol monohydrate
- Calcitonin
- Calcitonin Acetate
- Calcitonin salmon
- Calcitriol
- Calcium acetate
- Calcium ascorbate
- Calcium ascorbate dihydrate
- Calcium folinate
- Calcium fructoborate
- Calcium-glubionate
- Calcium hydrogenphosphate dihydrate
- Calcium lactate
- Calcium levofolinate
- Calcium Levulinate
- Calcium Pantothenate
- Calcium Pidolate
- Calcium polycarbophil
- Calcium Sennosides
- Camostat mesilate
- Camptothecin
- Camylofin
- Canagliflozin
- Canagliflozin hemihydrate
- Candesartan
- Cangrelor
- Canrenone
- Canthaxanthin
- Capecitabine
- Capecitabine Hydrochloride
- Capreomycin Sulfate
- Capsaicin
- Captopril
- Carbadox
- Carbaica
- Carbamazepine
- Carbamazepine Bromine
- Carbasalate calcium
- Carbazochrome
- Carbazochrome sodium sulfonate
- Carbenicillin disodium
- Carbetocin
- Carbidopa
- Carbimazole
- Carbinoxamine
- Carbinoxamine Maleate
- Carbocistein
- Carbocysteine
- Carbon disulphide
- Carboplatin
- Carboprost tromethamine
- Carboxymethylcellulose sodium salt
- Carfilzomib
- Carisoprodol
- Carnosic acid
- Caronic anhydride
- Carperitide
- Carpipramine
- Carprofen
- Carteolol
- Carteolol Hydrochloride
- Carvedilol
- Carvedilol hydrochloride
- Carvedilol Phosphate
- Casein
- Caspofungin
- Caspofungin acetate
- Cefaclor
- Cefadroxil
- Cefadroxil Monohydrate
- Cefalonium
- Cefalotin sodium
- Cefamandole nafate
- Cefathiamidine
- Cefatrizine
- Cefazolin
- Cefazolin sodium salt
- Cefcapene
- Cefcapene pivoxil
- Cefcapene pivoxil hydrochloride
- Cefdinir
- Cefditoren
- Cefditoren pivoxil
- Cefditoren sodium
- Cefepime
- Cefepime hydrochloride
- Cefetamet pivoxil hydrochloride
- Cefetamet pivoxyl
- Cefixime
- Cefixime Trihydrate
- Cefmenoxime hydrochloride
- Cefmetazole
- Cefmetazole Acid
- Cefmetazole sodium
- Cefminox sodium
- Cefodizime Acid
- Cefodizime sodium
- Cefonicid sodium
- Cefoperazone
- Cefoperazone sodium
- Cefoselis sulfate
- Cefotaxime
- Cefotaxime Sodium
- Cefotetan
- Cefotetan disodium
- Cefotiam
- Cefotiam Hexetil
- Cefotiam hydrochloride
- Cefoxitin
- Cefoxitin sodium
- Cefozopran
- Cefpimizole
- Cefpiramide sodium
- Cefpirome
- Cefpirome sulfate
- Cefpodoxime
- Cefpodoxime proxetil
- Cefprozil
- Cefquinome sulfate
- Cefradine
- Cefsulodin sodium
- Cefsulodine sodium
- Ceftazidime
- Ceftazidime dihydrochloride
- Ceftazidime Pentahydrate
- Cefteram
- Ceftezole
- Ceftezole sodium
- Ceftibuten
- Ceftibuten dihydrate
- Ceftiofur
- Ceftiofur hydrochloride
- Ceftiofur Sodium
- Ceftizoxime
- Ceftizoxime alapivoxil
- Ceftizoxime sodium
- Ceftriaxone
- Ceftriaxone disodium salt hemi(heptahydrate)
- Ceftriaxone sodium
- Cefuroxime Axetil
- Cefuroxime sodium
- Celecoxib
- Celiprolol hydrochloride
- Cephalexin
- Cephalomannine
- Cephalothin
- Cephapirin benzathine
- Cephaprin
- Cetilistat
- Cetirizine
- Cetirizine hydrochloride
- Cetrimide
- Cetrorelix
- Cetrorelix acetate
- Cetylpyridinium bromide
- Cetylpyridinium Chloride
- Cetylpyridinium chloride monohydrate
- Cevimeline
- Cevimeline Hydrochloride
- Chenodeoxycholic acid
- Chitosan
- Chlorambucil
- Chloramine B
- Chloranil
- Chlorantraniliprole
- Chlorcyclizine Hydrochloride
- Chlordiazepoxide
- Chlordiazepoxide hydrochloride
- Chlorhexidine acetate
- Chlorhexidine Base
- Chlorhexidine Gluconate
- Chlorhexidine hydrochloride
- Chlormadinone acetate
- Chloroacetone
- Chloroamphenicol stearate
- Chlorobutanol
- Chlorobutanol (anhydrous)
- CHLOROBUTANOL HEMIHYDRATE
- Chlorogenic acid
- Chloropyramine
- Chloropyramine hydrochloride
- Chloroquine Phosphate
- Chloroquine sulfate
- Chlorothiazide
- Chlorphenesin
- Chlorpheniramine
- Chlorpheniramine Maleate
- CHLORPHENIRAMINE TANNATE
- Chlorpromazine
- Chlorpromazine hydrochloride
- Chlorpropamide
- Chlorprothixene hydrochloride
- Chlortalidone
- Chlortetracycline
- Chlortetracycline hydrochloride
- Chlorzoxazone
- Cholecalciferol
- Cholesterol
- Choline bitartrate
- Choline chloride
- Choline Fenofibrate
- Choline glycerophosphate
- Choline Salicylate
- Choline Theophyllinate
- Chondroitin
- Chondroitin sulfate
- Chorionic gonadotrophin
- Chromium picolinate
- Chromium Polynicotinate
- Chymotrypsin
- Cibenzoline succinate
- Ciclesonide
- Ciclopirox
- Ciclopirox ethanolamine
- Ciclopirox olamine
- Cilansetron
- Cilastatin
- Cilastatin sodium
- Cilazapril
- Cilnidipine
- Cilostazol
- Cimetidine
- Cimetropium bromide
- Cinacalcet
- Cinacalcet Hydrochloride
- Cinchonidine
- Cinchonine
- Cinchophen
- Cinepazide Maleate
- Cinitapride Hydrogen Tartrate
- Cinnarizine
- Cinoxacin
- Ciprofibrate
- Ciprofloxacin
- Ciprofloxacin hydrochloride
- Ciprofloxacin Hydrochloride Hydrate
- Ciprofloxacin lactate
- Cisatracurium Besylate
- Cisplatin
- Citicoline Sodium
- Clarithromycin
- Clarithromycin Lactobionate
- Clemastine Fumarate
- Clevidipine Butyrate
- Cleviprex
- Clidinium Bromide
- Clindamycin
- Clindamycin Hydrochloride
- Clindamycin Palmitate Hydrochloride
- Clindamycin Phosphate
- Clinofibrate
- Clobazam
- Clobetasol Propionate
- Clobetasone Butyrate
- Clocapramine
- Clocortolone
- Clocortolone Pivalate
- Clofarabine
- Clofazimine
- Clomifene
- Clomifene Citrate
- Clomipramine
- Clomipramine Hydrochloride
- Clonidine
- Clonidine Hydrochloride
- Clopamide
- Cloperastine
- Cloperastine Hydrochloride
- Clopidogrel
- Clopidogrel Bisulfate
- Clopidogrel Hydrobromide
- Clopidogrel Hydrochloride
- Clopidogrel Hydrogen Sulfate
- Clopidol
- Cloprostenol Sodium
- Clorazepate Dipotassium
- Clorprenaline Hydrochloride
- Clorsulon
- Closantel Base
- Clotiazepam
- Clotrimazole
- Cloxacillin Benzathine
- Cloxacillin Sodium
- Cloxazolam .
- Clozapine
- Cobamamide
- Cobicistat
- Coenzyme
- Colchicine
- Colesevelam
- Colesevelam Hydrochloride
- Colestilan
- Colestipol hydrochloride
- Colistimethate Sodium
- Colistin
- Colistin Sulfate
- Collagen
- Conivaptan
- Conivaptan Hydrochloride
- Conjugated Estrogens
- Copovidone
- Copper Gluconate
- Cordycepin
- Cortisone
- Cortisone Acetate
- Coumarin
- Creatinine
- Crizotinib
- Crotamiton
- Curcumin
- Cyanocobalamin
- Cyanotemozolomide
- Cyclandelate
- Cyclizine
- Cyclizine Hydrochloride
- Cyclobenzaprine
- Cyclobenzaprine Hydrochloride
- Cyclopentene Oxide
- Cyclopentolate
- Cyclopentolate Hydrochloride
- Cyclophosphamide
- Cyclophosphamide Monohydrate
- Cycloserine
- Cyclosporin A
- Cyclosporine
- Cyfluthrin
- Cymiazole
- Cypermethrin
- Cyproheptadine
- Cyproheptadine Hydrochloride
- Cyproterone Acetate
- Cyromazine
- Cystine
- Cytarabine
- Cytarabine Hydrochloride
- Cytidylic Acid
- Cytisine
- Cytochrome C
- Cytosine
- (S)-(+)-2-Phenylpropionic Acid
- Dabrafenib Intermediate
- Dacarbazine
- Daclatasvir
- Daclatasvir Dihydrochloride
- Dactinomycin
- Dalbavancin
- Dalfampridine
- Dalfopristin
- Dalteparin
- Dalteparin Sodium
- D-Amphetamine Base
- D-Amphetamine Saccharate
- Danazol
- Danofloxacin Mesylate
- Dantrolene Sodium
- Dapagliflozin
- Dapsone
- Daptomycin
- Darifenacin
- Darifenacin Hydrobromide
- Darunavir
- DARUNAVIR AMORPHOUS
- Darunavir Ethanolate
- Dasatinib
- Dasatinib Monohydrate
- Daunorubicin
- Daunorubicin Hydrochloride
- D-Biotin
- D-Cloprostenol Sodium
- D-Cycloserine
- Deacetylnorgestimate
- Deacetyltaxol
- Decatol
- Decitabine
- Decloxizine
- Decloxizine Hydrochloride
- Decoquinate
- Deferasirox
- Deferiprone
- Deferoxamine Mesylate
- Deflazacort
- Degarelix
- Degarelix Acetate
- Dehydroepiandrosterone
- Dehydroepiandrosterone Enanthate
- Delafloxacin
- Delamanid
- Delavirdine
- Deltaline
- Deltamethrin
- Demeclocycline
- Demeclocycline Hydrochloride
- Denopamine
- Deoxyarbutin
- Deoxyribonuclease
- Depofemin
- Deprodone
- Dersalazine
- Descinolone
- Deserpidine
- Desflurane
- Desipramine Hydrochloride
- Desirudin
- Desloratadine
- Deslorelin
- Desmopressin
- Desmopressin Acetate
- Desogestrel
- Desonide
- Desoximetasone
- Desvenlafaxine
- Desvenlafaxine Fumarate
- Desvenlafaxine succinate
- Detomidine hydrochloride
- Dexamethasone
- Dexamethasone Acetate
- Dexamethasone Isonicotinate
- Dexamethasone Sodium Phosphate
- Dexamethasone Valerate
- Dexbrompheniramine Maleate
- Dexchlorpheniramine
- Dexchlorpheniramine Maleate
- Dexketoprofen trometamol
- Dexlansoprazole
- Dexmedetomidine
- Dexmedetomidine Hydrochloride
- Dexpramipexole
- Dexrabeprazole Sodium
- Dexrazoxane
- Dexrazoxane Hydrochloride
- Dextran
- Dextromethorphan Hydrobromide
- Dextrose Anhydrous
- Dextrose Monohydrate
- Dezocine
- D-Glucosamine Sulfate
- D-Glucuronolactone
- D-Glutamine
- Diacerein
- Diammonium Glycyrrhizinate
- Diatrizoate Meglumine
- Diatrizoate Sodium
- Diatrizoic Acid
- Diaveridine
- Diaveridine Hydrochloride
- Dibucaine Base
- Dibucaine Hydrochloride
- DI-Calcium Phosphate
- DICHLORALPHENAZONE
- Diclazuril
- Diclofenac
- Diclofenac Diethylamine
- Diclofenac Epolamine
- Diclofenac Potassium
- Diclofenac Sodium
- Dicloxacillin Sodium
- Dicyandiamide
- Dicyclomine Hydrochloride
- Dideoxyadenosine
- Dienogest
- Diethylamine Salicylate
- Diethylcarbamazine Citrate
- Difethialone
- Diflomotecan
- Diflorasone
- Diflorasone Diacetate
- Difloxacin Hydrochloride
- Diflucortolone
- Diflucortolone Valerate
- Diflunisal
- Difluprednate
- Digoxin
- Dihydralazine
- Dihydralazine Mesylate
- Dihydralazine Sulfate
- Dihydroartemisinin
- Dihydrocodeine Hydrogentartrate
- Dihydroergocristine Mesylate
- Dihydroergocryptine Mesylate
- Dihydroergotamine Mesylate
- Dihydroergotoxine Mesylate
- Dihydroquercetin
- Dihydrostreptomycin Sulfate
- Dihydroxyaluminum Sodium Carbonate
- Diloxanide Furoate
- Diltiazem
- Diltiazem hydrochloride
- Dimenhydrinate
- Dimercaptosuccinic acid
- Dimethyl fumarate
- Dimetridazole
- Diminazene
- Diminazene Diaceturate
- Diniconazole
- Dinitolmide
- Dinoprost Tromethamine
- Diosgenin
- Diosmetin
- Diosmin
- Diphemanil Mesylate
- Diphencyprone
- Diphenhydramine
- Industrial Grade Diphenhydramine Citrate
- Diphenhydramine Hydrochloride
- Diphenhydramine citrate
- Dipotassium Hydrogen Phosphate Trihydrate
- Diprophylline .
- Dipyridamole
- Diquafosol
- Dirithromycin
- Disodium Monofluorophosphate
- Disopyramide
- Disopyramide Phosphate
- Distigmine Bromide
- Disulfiram
- Dithranol
- Divalproex Sodium
- DL-alpha-Hydroxymethionine Calcium
- DL-Amphetamine Asphartate
- DL-Amphetamine Sulphate
- DL-Cloprostenol Sodium
- DL-Lysine
- DL-Lysine acetylsalicylate
- DL-Octopamine hydrochloride
- DL-O-Phosphorylserine
- DL-Selenomethionine
- D-Methyl Phenidate Hydrochloride
- D-Norpseudoe Phedrine Hydrochloride
- Dobutamine Hydrochloride
- Docetaxel
- Docetaxel anhydrous
- Docetaxel trihydrate
- Dofetilide
- Dolasetron mesylate
- Dolasteron
- Dolutegravir
- Dolutegravir Sodium
- Domiphen Bromide
- Domperidone
- Domperidone Maleate
- Donepezil Base
- Donepezil Hydrochloride
- Dopamine Hydrochloride
- Doramectin
- Doripenem
- Doripenem Monohydrate
- Dorzolamide
- Dorzolamide Hydrochloride
- Dothiepin
- Doxapram
- Doxapram Hydrochloride Monohydrate
- Doxazosin
- Doxazosin Mesylate
- Doxepin
- Doxepin Hydrochloride
- Doxercalciferol
- Doxibetasol
- Doxifluridine
- Doxofylline
- Doxorubicin
- Doxorubicin Hydrochloride
- Doxycycline
- Doxycycline Hyclate
- Doxycycline hydrochloride
- Doxycycline Monohydrate
- Doxylamine
- Doxylamine Succinate
- Drocinonide Phosphate Potassium
- Dromostanolone Propionate
- Dronabinol
- Dronedarone
- Dronedarone Hydrochloride
- Droperidol
- Drospirenone
- Drotaverine
- Drotaverine Hydrochloride
- Droxidopa
- Duloxetine
- Duloxetine Hydrochloride
- Dutogliptin
- Dybenal
- Dyclonine Hydrochloride
- Dynorphin A
- Ebastine
- Ebastine Fumarate
- Ecabet Sodium
- Econazole
- Econazole Nitrate
- Edaravone
- Edoxaban
- Edoxaban Tosylate
- Efavirenz
- EFINACONAZOLE
- Eflornithine
- Eflornithine Hydrochloride
- Eflornithine Hydrochloride Hydrate
- Efonidipine .
- Elastase
- Elcatonin
- Elcatonin Acetate
- Eldecalcitol
- Eledoisin
- Eletriptan
- Eletriptan Hydrobromide
- Eletriptan Hydrobromide Monohydrate
- Eliglustat
- Eluxadoline
- Elvitegravir
- Emedastine Difumarate
- Emetine Hydrochloride
- Empagliflozin
- Emtricitabine
- Enalapril
- Enalapril Maleate
- Enalaprilat
- Enfuvirtide
- Enkephalin
- Enoxacin
- Enoxaparin Sodium
- Enoxolone
- Enrofloxacin
- Enrofloxacin Hydrochloride
- Entacapone
- Entecavir
- Entecavir Hydrate
- Entecavir Monohydrate
- Enzalutamide
- Enzastaurin
- Epalrestat
- Eperisone Hydrochloride
- Epiandrosterone
- Epiandrosterone Acetate
- Epibrassinolide
- Epiestradiol
- Epinastine Hydrobromide
- Epinephrine
- Epinephrine Bitartrate
- Epirubicin
- Epirubicin Hydrochloride
- Epitiostanol
- Eplerenone
- Epocholeone
- Epoprostenol Sodium
- Epothilone B
- Eprazinone Dihydrochloride
- Eprinomectin
- Eprosartan
- Eprosartan Mesylate
- Eptaplatin
- Eptifibatide
- Eptifibatide Acetate
- Erdosteine
- Ergosterol
- Ergotamine Tartrate
- Eribulin
- Erlotinib
- Erlotinib Hydrochloride
- Ertapenem
- Ertapenem Disodium
- Ertapenem Sodium
- Erythritol
- Erythromycin
- Erythromycin Estolate
- Erythromycin Ethylsuccinate
- Erythromycin Lactobionate
- Erythromycin Oxime
- Erythromycin Phosphate
- Erythromycin Propionate
- Erythromycin Stearate
- Erythromycin Thiocyanate
- Eslicarbazepine
- Eslicarbazepine Acetate
- Esmolol Hydrochloride
- Esomeprazole
- Esomeprazole Magnesium
- Esomeprazole Magnesium Dihydrate
- Esomeprazole Magnesium Trihydrate
- Esomeprazole Sodium
- Estazolam
- Estradiol
- Estradiol Benzoate
- Estradiol Benzoate Butyrate
- Estradiol Cypionate
- Estradiol Dipropionate
- Estradiol Enanthate
- Estradiol Hemihydrate
- Estradiol Hexahydrobenzoate
- Estramustine Phosphate Sodium
- Estriol
- Estrone
- Eszopiclone
- Etafedrine Hydrochloride
- Etamiphylline Camsylate
- Etamsylate
- Etanercept
- Ethacridine Lactate
- Ethacrynate Sodium
- Ethacrynic Acid
- Ethanol
- Ethisterone
- Ethopabate
- Ethosuximide
- Ethyl Icosapentate
- Ethyl Loflazepate
- Ethyl Vanillin
- Ethylestrenol
- Ethylmorphine Hydrochloride
- Ethylparaben
- Ethynodiol Diacetate
- Ethynyl Estradiol
- Etidronate Disodium
- Etidronate Sodium
- Etiproston
- Etocrylene
- Etodolac
- Etofenamate
- Etofylline
- Etofylline Clofibrate
- Etofylline Nicotinate
- Etomidate
- Etonogestrel
- Etoposide
- Etoricoxib
- Etravirine
- Everolimus
- Exemestane
- Exenatide
- Exenatide Acetate
- Ezetimibe
- Ezogabine
- Faldaprevir
- Famciclovir
- Famotidine
- Faropenem Sodium
- Faropenem Sodium Hemipentahydrate
- Fasudil Hydrochloride
- Favipiravir
- Febantel
- Febuxostat
- Felbamate
- Felbinac
- Felodipine
- Felypressin
- Felypressin Acetate
- Fenazox
- Fenbendazole
- Fendizoic Acid
- Fenofibrate
- Fenofibric Acid
- Fenoprofen Calcium
- Fenoterol
- Fenoterol Hydrobromide
- Fenoxazoline Hydrochloride
- Fenpiverinium Bromide
- Fenproporex Hydrochloride
- Fenpyroximate
- Fenspiride
- Fenspiride Hydrochloride
- Fenticonazole Nitrate
- Ferric Citrate
- Ferrous Ascorbate
- Fertirelin
- Fertirelin Acetate
- Fesoterodine
- Fesoterodine Fumarate
- Fexofenadine
- Fexofenadine Hydrochloride
- Fidaxomicin
- Fimasartan
- Finasteride
- Fingolimod
- Fingolimod Hydrochloride
- Fipronil
- Flavopiridol Hydrochloride
- Flavoxate
- Flavoxate Hydrochloride
- Flecainide
- FLECAINIDE ACETATE
- Fleroxacin
- Flibanserin
- Flibanserin Hydrochloride
- Flofenicol
- Flomoxef
- Flonicamid
- Florfenicol
- Floxuridine
- Fluazuron
- Flubendazole
- Flucloxacillin Magnesium
- Flucloxacillin Sodium
- Fluconazole
- Fludarabine
- Fludarabine Phosphate
- Fludiazepam
- Fludioxonil
- Fludrocortisone
- Fludrocortisone Acetate
- Flufenamic Acid
- Flugestone
- Fluindione
- Flumequine
- Flumequine Sodium
- Flumethasone
- Flumethasone Acid
- Flumethrin
- Flumizole
- Flunarizine
- Flunarizine Dihydrochloride
- Flunisolide
- Flunixin Meglumine
- Fluocinolone Acetonide
- Fluocinonide
- Fluocortolone
- Fluorescein Sodium
- Fluorogesterone Acetate
- Fluorometholone
- Fluorometholone Acetate
- Fluoxetine
- Fluoxetine Hydrochloride
- Flupenthixol Dihydrochloride
- Fluphenazine Decanoate
- Fluphenazine Hydrochloride
- Flupirtine
- Flupirtine Maleate
- Fluprednidene
- Fluprednisolone
- FLUPREDNISOLONE ACETATE
- Flurazepam Dihydrochloride
- Flurbiprofen
- Flurbiprofen Axetil
- Flusilazole
- Flutamide
- Fluticasone
- Fluticasone Furoate
- Fluticasone Propionate
- Flutriafol
- Flutrimazole
- Fluvalinate
- Fluvastatin
- Fluvastatin Sodium
- Fluvoxamine
- Fluvoxamine Maleate
- Folcisteine
- Folic Acid
- Folinic Acid
- Follicle Stimulating Hormone
- Fomepizole
- Fondaparinux Sodium
- Formoterol Fumarate
- Formoterol Fumarate Dihydrate
- Forskolin
- Fosamprenavir Calcium
- Fosaprepitant
- Fosaprepitant Dimeglumine
- Foscarnet sodium
- Fosfluconazole
- Fosfomycin
- Fosfomycin Calcium
- Fosfomycin Phenylethylamine
- Fosfomycin Sodium
- Fosfomycin Trometamol
- Fosinopril
- Fosinopril Sodium
- Fosphenytoin Sodium
- Fozivudine
- Framycetin Sulphate
- Frovatriptan
- Frovatriptan Succinate
- Fudosteine
- Fulvestrant
- Fupentixol Dihydrochloride
- Furaltadone
- Furaltadone Hydrochloride
- Furazabol
- Furazolidone
- Furbenicillin
- Furosemide
- Fursultiamine
- Fursultimine Hydrochloride
- Fusidate Sodium
- Fusidine
- Gabapentin
- Gabapentin enacarbil
- Gabexate mesylate
- Gadobutrol
- Gadodiamide
- Gadofosveset Trisodium
- Gadopentetate Dimeglumine
- Gadoterate meglumine
- Galanthamine
- Galanthamine hydrobromide
- Gamithromycin
- Ganciclovir
- Ganciclovir Sodium
- Garenoxacin
- Garenoxacin Mesylate
- Gastrodin
- Gefitinib
- Geldanamycin
- Gemcitabine
- Gemcitabine hydrochloride
- Gemfibrozil
- Gemifloxacin
- Gemifloxacin Mesylate
- Gentamicin Sulfate
- Gestodene
- Gestonorone
- Gestrinone
- Gimeracil
- Glabridin
- Glatiramer Acetate
- Glibenclamide
- Gliclazide
- Glimepiride
- Glipizide
- Gliquidone
- Glucagon Hydrochloride
- Glucono Delta Lactone
- Glucosamine
- Glucosamine Hydrochloride
- Glucosamine Sulfate
- Glutaric Acid
- Glutathione
- Glycerol Phosphate Calcium Salt
- Glycine
- Glycopyrrolate
- Glycylglycine
- Glycyl-glycyl-glycine
- Glycyl-L-glutamine Monohydrate
- Glycyl-L-tyrosine
- Glycyrrhizic Acid Ammonium Salt
- Gly-Leu
- Gonadorelin
- Gonadorelin Acetate
- Goserelin
- Goserelin Acetate
- Gramine
- Granisetron Base
- Granisetron Hydrochloride
- Griseofulvin
- Growth Hormone Releasing Peptide (GHRP-6)
- Guaifenesin
- Guanethidine Sulfate
- Guanidine Nitrate
- Guanosine
- Halcinonide
- Halobetasol Propionate
- Halofantrine Hydrochloride
- Halofuginone
- Halometasone
- Haloperidol
- Haloperidol Decanoate
- Halquinol
- Heparin Sodium
- Heparinoid
- Heptaminol
- Heptaminol hydrochloride
- Hesperetin
- Hesperidin
- Hesperidin methylchalcone
- Hexaconazole
- Hexarelin
- Hexetidine
- Hexythiazox
- Histrelin
- Histrelin acetate
- Homatropine hydrobromide
- Homatropine methylbromide
- Homidium Chloride
- Hordenine
- Human GLP-1 (7-37)
- Huperzine A
- Hyaluronic acid
- Hyaluronidase
- Hydralazine
- Hydralazine hydrochloride
- Hydrazine acetate
- Hydrochlorothiazide
- Hydrocortisone
- Hydrocortisone aceponate
- Hydrocortisone acetate
- Hydrocortisone butyrate
- Hydrocortisone hemisuccinate
- Hydrocortisone sodium phosphate
- Hydrocortisone Sodium Succinate
- Hydrocortisone valerate
- Hydromadinone
- Hydroquinone
- Hydrotalcite
- Hydrotriamcinolone Acetate
- Hydroxocobalamin .
- Hydroxocobalamin acetate
- Hydroxocobalamin Hydrochloride
- Hydroxocobalamin Sulphate
- Hydroxyamphetamine Hydrobromide
- Hydroxychloroquine sulfate
- Hydroxyethyl Salicylate
- Hydroxyurea
- Hydroxyzine hydrochloride
- Hydroxyzine Pamoate
- Hygromycin B
- Hyoscine Butylbromide
- Hyoscyamine sulphate
- Hyoscyamine sulphate hydrate
- Hypoxanthine
- Ibandronate Sodium
- Ibudilast
- Ibuprofen
- Ibuprofen Arginine
- Ibutilide fumarate
- Icatibant
- Icatibant acetate
- Idarubicin
- Idarubicin hydrochloride
- Idebenone
- Idelalisib
- Ifenprodil
- Ifenprodil tartrate
- Ifosfamide
- Ilaprazole
- Iloperidone
- Iloprost
- Imatinib mesylate
- Imazamox
- Imidacloprid
- Imidafenacin
- Imidapril
- Imidapril Hydrochloride
- Imidazole
- Imidocarb dipropionate
- Imipenem
- Imipramine hydrochloride
- IMIPRAMINE PAMOATE
- Imiquimod
- Indacaterol
- Indacaterol maleate
- Indapamide
- Indinavir
- Indinavir sulfate
- Indometacin
- Indomethacin
- Indomethacin sodium
- Infliximab
- Ingenol Mebutate
- Inosine
- Inositol
- Intedanib
- Iodixanol
- Iohexol
- Iopamidol
- Iotrolan
- Ioversol
- Ipragliflozin
- Ipragliflozin L-Proline
- Ipratropium Bromide
- Ipriflavone
- Irbesartan
- Irinotecan
- Irinotecan hydrochloride
- Irinotecan hydrochloride trihydrate
- Iron lll Hydroxide Poly Maltose complex
- Iron sorbitol
- Iron Sucrose
- Iron-dextran
- Irsogladine
- Isavuconazole
- Isavuconazonium sulfate
- Isepamicin sulfate
- Isocarboxazid
- Isoconazole nitrate
- Isoflavone
- Isoflupredone
- Isoflupredone acetate
- Isoflurane
- Isometamidium chloride
- Isometamidium Chloride Hydrochloride
- Isometheptene mucate
- Isoniazid
- Isophosphamide
- Isoprenaline hydrochloride
- Isoprenaline sulphate
- Isoprinosine
- Isopropamide Bromide
- Isopropamide iodide
- Isopropyl Methylphenol
- Isopropyl unoprostone
- Isoproterenol
- Isosorbide 5-mononitrate
- Isosorbide dinitrate
- Isosulfan Blue
- Isothipendyl
- Isotretinoin
- Isoxadifen ethyl ester
- Isoxaflutole
- Isoxsuprine hydrochloride
- Istradefylline
- Itopride
- Itopride hydrochloride
- Itraconazole
- Ivabradine
- Ivabradine hydrochloride
- Ivacaftor
- Ivermectin
- Ixabepilone
- Josamycin
- Julolidine
- Kanamycin
- Kanamycin sulfate
- Ketazolam .
- Ketoconazole
- Ketoketal
- Ketoprofen
- Ketoprofen lysinate salt
- Ketorolac
- Ketorolac Tromethamine
- Ketotifen
- Ketotifen Fumarate
- Kitasamycin
- Kitasamycin tartrate
- Kojic acid
- L(-)-Glutathione
- L(+)-Ascorbic acid
- L-(+)-Glutamic acid hydrochloride
- LABETALOL
- Labetalol Hydrochloride
- Labetalone hydrochloride
- Lacidipine
- Lacosamide
- Lactic Acid
- Lactose
- Lafutidine
- Lagociclovir .
- Lamivudine
- Lamivudine Salicylate
- Lamotrigine
- Lanoconazole
- Lanolin
- Lanreotide
- Lanreotide acetate
- Nutraceuticals .
- Chemical Intermediates
- (1R,2R) 1,2 Cyclohexanedimethanol
- N Lodosuccinimide
- Pharmaceutical excipients
- (R)-(+) Glycidol
- (1alpha)-17-(Acetyloxy)-6-chloro-1-(chloromethyl)
- (1R,2R,3S,5R) (-) 2,3 Pinanediol
- (2-Fluorobenzyl) Hydrazine
- (3,4-Dimethoxyphenyl) Acetic Acid
- (3R) 3 Amino 1 Butanol
- Bromomethyl Cyclopropane
- (Phenylazo) Malonitrile
- (1-Benzyl-4-piperidyl)methanol
- (1-Ethyl-2-Phenoxy-Propyl)- Hydrazine L Dibenzoly
- (1-Hydroxycyclohexyl)(4-hydroxyphenyl)acetonitril
- (1-Hydroxycyclopentyl)Phenylacetic Acid
- (1-Methyl-4-piperidinyl)[3-[2-(3-chlorophenyl)eth
- (1-Methylpiperidin-4-Yl) 2-Hydroxy-2,2-Di(Phenyl)
- (1R,1'R)-2,2'-(3,11-Dioxo-4,10-dioxatridecamethyl
- (1R,2R,3S,5R)-(-)-2,3-Pinanediol
- (1R,2R)-1-[(3,4-Dimethoxyphenyl)methyl]-2-[3-(ter
- (1R,2R)-1,2-Cyclohexanedicarboxylic acid
- (1R,2R)-1,2-Cyclohexanedimethanol
- (1R,2R)-2-(3,4-difluorophenyl)cyclopropanecarboxy
- (1R,2R)-2-Amino-1-(4-nitrophenyl)propane-1,3-diol
- (1R,2R)-(-)-1,2-Diaminocyclohexane
- (1R,2R)-(+)-1,2-Diphenylethylenediamine
- (1R,2R)-(-)-N-(4-Toluenesulfonyl)-1,2-diphenyleth
- (1R,2S,5R)-7-Oxo-6-(phenylmethoxy)-1,6-diazabicyc
- (1R,2S,5R)-Menthyl-(2R,5S)-5-(4-amino-2-oxo-2H-py
- (1R,2S)-1-Amino-2-ethenylcyclopropanecarboxylic a
- (1R,2S)-1-Amino-2-vinylcyclopropanecarboxylic aci
- (1R,2S)-1-Amino-N-(cyclopropylsulfonyl)-2-ethenyl
- (1R,2S)-1-tert-Butoxycarbonylamino-2-vinylcyclopr
- (1R,2S)-2-(3,4-Difluorophenyl)cyclopropanamine
- (1R,2S)-2-(3,4-Difluorophenyl)cyclopropanamine (2
- (1R,2S)-2-(3,4-Difluorophenyl)cyclopropanaminium
- (1R,2S)-2-Amino-1,2-diphenylethanol
- (1R,2S)-2-Ethenyl-1-[[[(1R,2R,4S)-2-[(5-hexen-1-y
- (1R,2S)-2-Fluorocyclopropylamine tosylate
- (1R,2S)-2-[(Phenylmethoxy)methyl]-3-cyclopenten-1
- (1R,2S)-Ethyl 1-amino-2-vinylcyclopropanecarboxyl
- (1R,2S)-Methyl 1-amino-2-vinylcyclopropanecarboxy
- (1R,2S)-rel-2-(3,4-Difluorophenyl)cyclopropanamin
- (1R,3aR,7aR)-1-((R)-6-hydroxy-6-Methylheptan-2-yl
- (1R)-2-[[6-[2-[(2,6-Dichlorobenzyl)oxy]ethoxy]hex
- (1R,3aR,7aR)-1-((S)-1-hydroxypropan-2-yl)-7a-Meth
- (1R)-(-)-Menthyl glyoxylate hydrate
- (10-Phenylanthracen-9-yl)boronic acid
- (17beta)-13-Ethyl-17-hydroxy-11-methylenegon-4-en-
- (17)-3,3-[1,2-Ethanediylbis(oxy)]-17-hydroxy-19-
- (-)-1-[(4-Chlorophenyl)phenylmethyl]piperazine
- (-)-4-(4-Dimethylamino)-1-(4-fluorophenyl)-1-(hyd
- ((5-Thiazolyl)methyl)-(4-nitrophenyl)carbonate
- (+)-2,3-O-Isopropylidene-L-threitol
- (+)-5-Iodo-2'-deoxyuridine
- (+)-(3aR,4R,5r,6aS)-Hexahydro-5-hydroxy-4-[(1E,3R
- (+)-(6R)-2,6-Diamino-4,5,6,7-tetrahydrobenzothiaz
- (+%2f-)1-(1-Naphthyl)ethylamine
- (+%2f-)-2,2-Dimethyl-1,3-dioxolane-4-methanol
- (+%2f-)-4-[4-(diMethylaMino)-1-(4-fluorophenyl)-1
- (+%2f-)-trans-1,2-Diaminocyclohexane
- ()-1,2-Diphenyl-1-[4-[2-(dimethylamino)ethoxy]ph
- (1 R)-3-Chloro-1-phenyl-propan-1-ol
- (1R,2R,4R)-2-[(5-Hexen-1-yl methylamino)carbonyl]-
- (1R,2R,4S)-2-[(5-Hexen-1-yl methylamino)carbonyl]-
- (1R,2S)-1-[[(1,1-Dimethylethoxy)carbonyl]amino]-2-
- (1R,2S)-2-Amino-1-(4-hydroxyphenyl)propan-1-ol
- (1 R)-N-Benzyl-1-(1-Naphthyl)Ethylamine Hydrochlor
- (-)-4-Chlorobenzhydrylamine
- (-)-Corey aldehyde benzoate
- (-)-Corey lactone benzoate
- (-)-Corey lactone 4-phenyl benzoate alcohol
- (-)-Corey lactone diol
- (+)-Di-1,4-toluoyl-D-tartaric acid
- (+)-Dibenzoyl-D-tartaric acid
- (+)-Dibenzoyl-D-tartaric acid monohydrate
- (-)-Dibenzoyl-L-tartaric acid monohydrate
- (-)-Diethyl D-tartrate
- (-)-Diisopinocampheyl chloroborane
- (+)-Diisopinocampheyl chloroborane
- (-)-Di-p-toluoyl-L-tartaric acid
- (+)-Methyl (S)-3-hydroxyvalerate
- (-)-O-Acetyl-D-mandelic acid
- (+)-O,O-Di-p-toluoyl-D-Tartaric acid Monohydrate
- (1R-2R)-2-(3,4-diflrorophenyl)cyclopropanamine(2R)
- (1R-2R)-2-(3,4-diflrorophenyl)cyclopropanamine
- (1R-2R)-2-(3,4-diflrorophenyl)cyclopropanamine(R)-
- (1R-2R)-2-(diflrorophenyl)cyclopropanecarboxamide
- (1R-2R)-2-(diflrorophenyl)cyclopropanamine(2R,3R)-
- (1R,3aR,7aR)-1-((S)-1-hydroxypropan-2-yl)-7a-Methy
- (1R,3R)-1-(1,3-Benzodioxol-5-yl)-2-(chloroacetyl)-
- (1R,3S,4S)-3-(6-Bromo-1H-benzimidazol-2-yl)-2-azab
- (1R,3s,5S)-3-(3-Isopropyl-5-methyl-4H-1,2,4-triazo
- (1R,3S,4S)-3-[6-(4,4,5,5-Tetramethyl-1,3,2-dioxabo
- (1R,3S,4S)-N-Boc-2-azabicyclo[2.2.1]heptane-3-carb
- (1R,4R,5R)-3-Oxo-2-oxabicyclo[2.2.1]heptane-5-carb
- (1R,4S)-2-Azabicyclo[2.2.1]hept-5-en-3-one
- (1R,4S)-4-Aminocyclopentene-1-methanol hydrochlori
- (1R,5S)-5-(Dimethylphenylsilyl)-2-(hydroxymethyl)-
- (1R,6R)-2,8-Diazabicyclo[4,3,0]nonane
- (1S,2S,3R,4R)-methyl 3-((R)-1-acetamido-2-ethylbut
- (1S,2S,3R,5S)-(+)-2,3-Pinanediol
- (1S,2S,3S,4R)-3-[(1S)-1-Amino-2-ethyl butyl]-4-[[(
- (1S,2S,3S,5S)-5-(2-Amino-6-(benzyloxy)-9H-purin-9-
- (1S,2S,3S,5S)-5-[2-[[(4-Methoxyphenyl)diphenylmeth
- (1S,2S)-(1-Benzyl-3-chloro-2-hydroxypropyl)carbami
- (1S,2S)-(-)-1,2-Diphenyl-1,2-ethanediamine
- (1S,3aR,6aS)-(2S)-2-Cyclohexyl-N-(2-pyrazinylcarbo
- (1S,3aR,6aS)-Octahydrocyclopenta[c]pyrrole-1-carbo
- (1S,3S,5S)-2-Azabicyclo[3.1.0]hexane-3-carboxamide
- (1S,3S,5S)-2-Azabicyclo[3,3,0]octane-3-carboxylic
- (1S,4R)-4-(2-Amino-6-chloro-9H-purin-9-yl)-2-cyclo
- (1S,4R)-4-[[(1,1-Dimethylethoxy)carbonyl]amino]-2-
- (1S,4R)-4-Amino-2-cyclopentene-1-carboxylic Acid M
- (1S,4R)-(4-Aminocyclopent-2-enyl)methanol hydrochl
- (1S,4R)-cis-4-Acetoxy-2-cyclopenten-1-ol
- (1S,5R,6S)-Ethyl 5-(pentan-3-yl-oxy)-7-oxa-bicyclo
- (1S,5R)-1-Phenyl-3-oxabicyclo[3.1.0]hexan-2-one
- (1S)-1,5-Anhydro-1-C-[3-(benzo[b]thien-2-ylmethyl)
- (1S)-1,5-Anhydro-1-C-[4-chloro-3-[(4-ethoxyphenyl)
- (1S)-1-Phenyl-1,2,3,4-tetrahydroisoquinoline
- (1S)-4,5-dimethoxy-1-aminomethyl-benzocyclobutane
- (1S)-4,5-Dimethoxy-1-[(methylamino)methyl]benzocyc
- (1S-cis)-4-Amino-2-cyclopentene-1-methanol
- (1S-cis)-4-Amino-2-cyclopentene-1-methanol D-hydro
- (2,2-Dimethoxyethoxy)-Benzene
- (2b,3a,5a,16b,17b)-2-(4-Morpholinyl)-16-(1-pyrroli
- (2b,3a,5a,16b,17b)-17-Acetoxy-3-hydroxy-2-(4-morph
- (2-Bromo-1,1-difluoroethyl)benzene
- (2-Chlorophenyl)Acetaldehyde
- (2E,4R)-5-[1,1'-Biphenyl]-4-yl-4-[[(1,1-dimethylet
- (2E)-3-(5-Nitro-1-cyclohexen-1-yl)-2-propenoic aci
- (2E)-3-[(4S)-2,2-Dimethyl-1,3-dioxolan-4-yl]-2-met
- (2-Fluorobenzyl)hydrazine
- (2-Formamido-1,3-thiazol-4-yl)glyoxylic acid
- (2-Methyl-2-Phenyl-Propyl) Acetate
- (2-Methyl-5-nitrophenyl)guanidine nitrate
- (2r,3s)-benzyloxycarbonylamino no-2-hydroxy-1-(N-i
- (2'R)-2'-Deoxy-2'-fluoro-2'-methyluridine
- (2'R)-2'-Deoxy-2'-fluoro-2'-methyluridine 3',5'-di
- (2R,3R)-1-(Dimethylamino)-3-(3-methoxyphenyl)-2-me
- (2R,3R)-2-(2,4-difluorophenyl)-3-(tetrahydro-2H-py
- (2R,3R)-2-(2,4-Difluorophenyl)-1-(1H-1,2,4-triazol
- (2R,3R)-2,3-Bis[(4-methylbenzoyl)oxy]butanedioic a
- (2R,3S)-2-(2,4-Difluorophenyl)-3-methyl-[(1H-1,2,4
- (2R,3S)-2-(4-Ethyl-2,3-dioxo-1-piperazinecarboxami
- (2R,3S)-2-[(1R)-1-[3,5-Bis(trifluoromethyl)phenyl]
- (2R,3S)-3-(tert-Butoxycarbonyl)amino-1,2-epoxy-4-p
- (2R)-1-(2,4-Difluorophenyl)-2-[(tetrahydro-2H-pyra
- (2R)-2-[(4-Ethyl-2,3-dioxopiperazinyl)carbonylamin
- (2R)-2-Deoxy-2-fluoro-2-methyl-D-erythropentonic a
- (2R)-4-Benzyl-2-[(1R)-1-[3,5-bis(trifluoromethyl)p
- (2'R)-N-Benzoyl-2'-deoxy-2'-fluoro-2'-methylcytidi
- (2R)-rel-6-Fluoro-3,4-dihydro-2-(2R)-2-oxiranyl-2H
- (2R)-rel-6-Fluoro-3,4-dihydro-2-[(2S)-2-oxiranyl]-
- (2R,3S 2S,3R)-2-(2,4-Difluorophenyl)-3-(5-fluoro-4
- (2R,3S,4R,5R)-2-(hydroxyMethyl)-5-(6-((2-(Methylth
- (2R,4R)-1-[(2S)-5-[[Imino(nitroamino)methyl]amino]
- (2R,4S,5R)-3-Benzoyl-2-(4-methoxyphenyl)-4-phenyl-
- (2R,4S)-4-Amino-5-(biphenyl-4-yl)-2-methylpentanoi
- (2R,4S)-5-(Biphenyl-4-yl)-4-[(tert-butoxycarbonyl)
- (2R,5R)-1,6-Diphenyl-2,5-hexanediamine
- (2R,5R)-1,6-Diphenyl-2,5-hexanediamine hydrochlori
- (2R,5R)-5-Hydroxy-1,3-oxathiolane-2-carboxylic aci
- (2R-cis)-4-Amino-1-[2-(hydroxymethyl)-1,3-oxathiol
- (2S,2'S)-2,2'-([1,1'-Biphenyl]-4,4'-diyldi-1H-imid
- (2S,3aR,7aS)-1H-Octahydroindole-2-carboxylic acid
- (2S,3R)-3-((4R)-2,2-Dimethyldioxolan-4-yl)-2-methy
- (2S,3S,5S)-2-(2,6-Dimethylphenoxyacetyl)amino-3-hy
- (2S,3S,5S)-2-(N-((5-Thiazolyl)methoxyca-rbonyl)ami
- (2S,3S,5S)-5-Amino-2-(N-((5-thiazolyl)-methoxycarb
- (2S,3S,5S)-5-(tert-Butoxycarbonylamino)-2-(N-5-thi
- (2S,3S,5S)-5-tert-Butoxycarbonylamino-2-amino-3-hy
- (2S,3S)-1,2-Epoxy-3-(Boc-amino)-4-phenylbutane
- (2S,3S)-1,2-Epoxy-3-(Cbz-amino)-4-phenylbutane
- (2S,3S)-2-(2,4-Difluorophenyl)-1-(1H-1,2,4-triazol
- (2S,3S)-3-aMino-1-chloro-4-phenylbutan-2-ol hydroc
- (2S,3S)-3-Amino-N-cyclopropyl-2-hydroxyhexanamide
- (2S,4E)-5-Chloro-N,N-dimethyl-2-(1-methylethyl)-4-
- (2S,4R)-2-(Hydroxymethyl)-4-[(methylsulfonyl)oxy]-
- (2S,4S)-1-(tert-butoxycarbonyl)-4-(MethoxyMethyl)p
- (2S,4S)-4-(Acetylthio)-2-(hydroxymethyl)-1-pyrroli
- (2S,4S)-4-Cyano-1,2-pyrrolidinedicarboxylic acid 1
- (2S,4S)-4-(Methoxymethyl)-1,2-pyrrolidinedicarboxy
- (2S,5R)-5-[(Phenylmethoxy)amino]-2-piperidinecarbo
- (2S,5S)-1-(4-(tert-butyl)phenyl)-2,5-bis(4-nitroph
- (2S,5S)-5-methylpyrrolidine-2-carboxylic acid
- (2S,5S)-N-Boc-5-methylpyrrolidine-2-carboxylic aci
- (2S,6R)-6-Amino-2-(2-thienyl)-1,4-thiazepan-5-one
- (2S)-1-[[(2-Amino-1,1-dimethylethyl)amino]acetyl]-
- (2S)-1-(Chloroacetyl)-2-pyrrolidine carbonitrile
- (2S)-1-Methyl-2-pyrrolidone carboxylic acid
- (2S)-2-(2-Oxopyrrolidin-1-yl)butanoic acid
- (2S)-2-Amino-4-methyl-1-[(2R)-2-methyloxiranyl]-1-
- (2S)-2-Cyclohexyl-N-(2-pyrazinylcarbonyl)glycyl-3-
- (2S)-3-Amino-1,2-Propanediol
- (2S)-4-(1,3-Dioxoisoindolin-2-yl)-2-hydroxybutanoi
- (2S)-4-Oxo-2-(3-thiazolidinylcarbonyl)-1-pyrrolidi
- (2S)-5-[(2,5-Dioxo-1-pyrrolidinyl)oxy]-5-oxo-2-[(1
- (2S)-(1-Tetrahydropyramid-2-one)-3-methylbutanoic
- (2S-cis)-2-[(Dimethylamino)carbonyl]-4-mercapto-1-
- (2S-cis)-(+)-2,3-Dihydro-3-hydroxy-2-(4-methoxyphe
- (2S)-Hydroxy(Phenyl)Acetic Acid (1S)-3-(Dimethylam
- (2S)-N-(2,6-Dimethylphenyl)-2-piperidinecarboxamid
- (2S-trans)-3-Amino-2-methyl-4-oxoazetidine-1-sulph
- (2Z)-4-Oxo-4-[3-(trifluoromethyl)-5,6-dihydro-[1,2
- (3-(2-Aminoethyl)-1H-indol-5-yl)-N-methyl methanes
- (3-[5-Cyano-1-(4-fluorophenyl) (1,3-dihydroisobenz
- (3,4-Dimethoxyphenyl)acetic acid
- (3,4-Dimethoxyphenyl)acetonitrile
- (3,5-Diphenylphenyl)boronic acid
- (3alpha,5beta,6E)-6-Ethylidene-3-hydroxy-7-oxochol
- (3alpha,5beta)-6-Ethylidene-3-hydroxy-7-oxocholan-
- (3-Amino-Pyridin-2-Yl)-Methanol
- (3aR,4R,5R,6aS)-5-(Benzoyloxy)hexahydro-4-[(1E)-3-
- (3aR,4R,5R,6aS)-hexahydro-5-triethylsilane-4-((E)-
- (3aR,4R,5R,6aS)-Hexahydro-4-[(1E,3S)-3-hydroxy-5-p
- (3aR,4R,6S,6aS)-4-[[(1,1-Dimethylethoxy)carbonyl]a
- (3aR,4R,6S,6aS)-4-(tert-butoxycarbonylaMino)-3-(pe
- (3aR,4S,6R,6aS)-6-[7-Amino-5-(propylthio)-3H-1,2,3
- (3aR,4S,6R,6aS)-6-Aminotetrahydro-2,2-dimethyl-4H-
- (3aR,4S,7R,7aS)-rel-Hexahydro-4,7-methano-1H-isoin
- (3aR,7aR)-4'-(1,2-Benzisothiazol-3-yl)octahydrospi
- (3aR,12bR)-rel-5-Chloro-2,3,3a,12b-tetrahydro-2-me
- (3aR,4S,6R,6aS)-6-aminotetrahydro-2,2-dim
- (3aS,4R,5S,6aR)-(+)-Hexahydro-5-hydroxy-4-(hydroxy
- (3aS,4R,6S,6aR)-(6-Amino-2,2-dimethyl-tetrahydrocy
- (3aS,4S,6aR)-Tetrahydro-4-methoxy-furo[3,4-b]furan
- (3aS,6aR,9aR,9bS)-Decahydro-3,6,9-tris(methylene)a
- (3aS,6aR)-Tetrahydro-4-methoxyfuro[3,4-b]furan-2(3
- (3b,5b,15a,16a)-15,16-Dihydro-3,5-dihydroxy-3'H-cy
- (3beta,7alpha,15alpha)-3,7,15-Trihydroxy-androst-5
- (3-Chloropropyl)carbamic acid tert-butyl ester
- (3-Chloropropyl)pyrrolidine hydrochloride
- (3-Dimethylamino)propyltriphenylphosphonium bromid
- (3E)-2,3-Dihydro-3-(methoxyphenylmethylene)-2-oxo-
- (3-Fluoro-4-morpholin-4-ylphenyl)carbamic acid ben
- (3-Fluoro-4'-Pentyl-4-Biphenylyl)Boronic Acid
- (3R,3aR,4S,4aR,7R,8aR,9aR)-7-[(Ethoxycarbonyl)amin
- (3R,3aS,4S,4aS,7R,9aR)-1,3,3a,4,4a,5,6,7,8,9a-Deca
- (3R,3aS,6aR)-Hexahydrofuro[2,3-b]furan-3-ol
- (3R,4R)-1-Benzyl-N,4-dimethylpiperidin-3-amine
- (3R,4R)-3,4-Dimethyl-4-(3-hydroxyphenyl)piperidine
- (3R,4R)-N,4-Dimethyl-1-(phenyl methyl)-3-piperidin
- (3R,4R)-N,4-Dimethyl-1-(phenylmethyl)-3-piperidina
- (3R,4S)-1-(4-Fluorophenyl)-3-[(3S)-3-(4-fluorophen
- (3R,4S)-1-Benzoyl-3-(1-ethoxyethoxy)-4-phenyl-2-az
- (3R,4S)-1-Benzoyl-4-phenyl-3-[(triethylsilyl)oxy]-
- (3R,4S)-3-Hydroxy-4-phenyl-2-azetidinone
- (3R,4S)-4-[4-(Benzyloxy)phenyl]-1-(4-fluorophenyl)
- (3R,4S)-4-Phenyl-3-[(Triethylsilyl)Oxy]-2-Azetidin
- (3R,4S)-tert-Butyl 2-oxo-4-phenyl-3-(triethylsilyl
- (3R,5S,6E)-7-[2-Cyclopropyl-4-(4-fluorophenyl)-3-q
- (3R,5S,6E)-7-[4-(4-fluorophenyl)-6-isopropyl-2-(N-
- (3R,5S)-7-[2-Cyclopropyl-4-(4-fluorophenyl)-3-quin
- (3R)-3-[4-[(5-Bromo-2-chlorophenyl)methyl]phenoxy]
- (3R)-3-Amino-1-butanol
- (3R)-3-(tert-Butyldimethylsilyl)oxypentanedioate-1
- (3R)-5-Oxo-3-[(triethylsilyl)oxy]-1-cyclopentene-1
- (3S,4a,8aS)-2-[(2R,3S)-3-Amino-2-hydroxy-4-phenylb
- (3S,4R)-3-[(1R)-1-Hydroxyethyl]-4-[(1R)-1-methyl-3
- (3S,4R)-4-(4-Fluorophenyl)-3-hydroxymethyl-1-methy
- (3S,4R)-4-Acetoxy-3-[(R)-1-(tert-butyldimethylsily
- (3S,4S)-3-((R)-(tert-Butyldimethyl-silyloxy)ethyl)
- (3S,4S)-N,4-Dimethyl-1-(phenylmethyl)-3-piperidina
- (3S,5S)-3-(1-Methylethyl)-2-oxo-5-[(2S,4S)-tetrahy
- (3S)-3-[4-[(2-Chloro-5-iodophenyl)methyl]phenoxy]t
- (3S,5S)-5-[(1R,3S)-1-Bromo-3-[[4-methoxy-3-(3-meth
- (3S)-3-[4-[(5-Bromo-2-chlorophenyl)methyl]phenoxy]
- (3S)-3-Amino-2,3,4,5-Tetrahydro-2-Oxo-1H-1-Benzaze
- (3S)-3-Amino-N-cyclopropyl-2-hydroxyhexanamide hyd
- (3S)-3-(tert-Butoxycarbonyl)amino-1-chloro-4-pheny
- (3S)-Tetrahydro-3-Furanyl 4-Methylbenzenesulfonate
- (3Z)-2,3-Dihydro-3-[[[4-[methyl[2-(4-methyl-1-pipe
- (4-[1,2,4] Triazol-1-yl methyl phenyl) hydrazine
- (4-Amino-2-nitrophenyl)carbamic acid ethyl ester
- (4aR,4bS,6aS,7S,9aS,9bS)-2,3,4,4a,4b,5,6,6a,7,8,9,
- (4aR,7aR)-Octahydro-1H-Pyrrolo[3,4-b]Pyridine
- (4aS,7aS)-Octahydro-6-(Phenylmethyl)-1H-Pyrrolo[3,
- (4-Chloro-2-Ethoxycarbonyl)Benzeneboronic Acid
- (4-Chlorophenoxy)acetyl chloride
- (4-Nitrophenyl)methanesulfonyl chloride
- (4-Nitrophenyl)methyl 3-oxobutanoate
- (4R,6R)-6-[2-[2-(4-fluorophenyl)-5-(1-methylethyl)
- (4R,6R)-tert-Butyl-6-(2-aminoethyl)-2,2-dimethyl-1
- (4R,6R)-tert-Butyl-6-cyanomethyl-2,2-dimethyl-1,3-
- (4R,6S)-6-[(1E)-2-[2-Cyclopropyl-4-(4-fluorophenyl
- (4R,12aS)-3,4,6,8,12,12a-Hexahydro-7-hydroxy-4-met
- (4R,12aS)-3,4,6,8,12,12a-Hexahydro-7-methoxy-4-met
- (4R,12aS)-N-[(2,4-Difluorophenyl)methyl]-3,4,6,8,1
- (4R)-4-[[(1,1-Dimethylethyl)Dimethylsilyl]Oxy]-2-C
- (4R)-4-Hydroxy-N,N-diphenyl-2-pentynamide
- (4R)-5-[1,1'-Biphenyl]-4-yl-4-[[(1,1-dimethylethox
- (4R-Cis)-6-[(Acetyloxy) methyl]-2,2-Dimethyl-1,3-D
- (4R-Cis)-6-Hydroxymethyl -2,2-Dimethyl-1,3-Dioxane
- (4R-Cis)-6-Hydroxymethyl-2,2-dimethyl-1,3-dioxane-
- (4S,4aS,5aR,12aS)-9-Amino-4,7-bis(dimethylamino)-1
- (4S,5R)-3-(tert-Butoxycarbonyl)-2,2-dimethyl-4-phe
- (4S,5R)-5-[3,5-Bis(trifluoromethyl)phenyl]-4-methy
- (4S,6S)-4H-Thieno[2,3-b]-thiopyran-4-ol-5,6-dihydr
- (4S,6S)-5,6-Dihydro-4-hydroxy-6-methylthieno[2,3-b
- (4S)-3-[5-(4-Fluorophenyl)-1,5-dioxopenyl]-4-pheny
- (4S)-3-[(2S,4S)-4-Azido-2-(1-methylethyl)-1-oxo-4-
- (4S)-3-[(5S)-5-(4-Fluorophenyl)-5-hydroxypentanoyl
- (4S)-4-Acetamide-5,6-Dihydro-6-Methyl-2-Sulfonamid
- (4S)-(-)-4-Isopropyl-2-oxazolidinone
- (4S-trans) 4H-thieno[2.3-b] thiopyran-4-ol,5,6-dih
- (5a,11b,17b)-11-[4-(Dimethylamino)phenyl]-5,17-dih
- (5alpha,10alpha)-5,10-Epoxy-17-hydroxy-19-norpregn
- (5alpha,11beta)-11-[4-(Dimethylamino)phenyl]-5,17-
- (5-Amino-2-butyl-3-benzofuranyl)[4-[2-(dibutylamin
- (5-Amino-2-butyl-3-benzofuranyl)[4-[3-(dibutylamin
- (5-bromo-2-chlorophenyl)(4-hydroxyphenyl)methanone
- (5-Bromo-2-chlorophenyl)(4-ethoxyphenyl)methanone
- (5-Bromo-2-chlorophenyl)(4-methoxyphenyl)-methanon
- (5R)-3-(3-Fluoro-4-(4-morpholinyl)phenyl)-5-hydrox
- (5R)-3-(4-Bromo-3-fluorophenyl)-5-hydroxymethyloxa
- (5R)-4-(5-Chloro-1,3-benzoxazol-2-yl)-5-methyl-1,4
- (5R)-5-(2,2-Dimethyl-4H-1,3-benzodioxin-6-yl)-1,3-
- (5R-cis)-Toluene-4-sulfonic acid 5-(2,4-difluoroph
- (5S,6E)-7-[4-(4-Fluorophenyl)-6-(1-methylethyl)-2-
- (5S)-2-Oxo-1,5-imidazolidinedicarboxylic acid 5-(1
- (5S)-3-Methyl-2-oxo-1,5-imidazolidinedicarboxylic
- (5S)-N-(Methoxycarbonyl)-L-valyl-5-methyl-L-prolin
- (5Z)-N-(Cyclopropylmethyl)-7-[(1R,2R,3R,5S)-3,5-di
- (6a,11b,16a,17a)-6,9-Difluoro-11,17-dihydroxy-16-m
- (6a,11b,16a,17a)-6,9-Difluoro-11-hydroxy-16-methyl
- (6-Chloro-3-pyridinyl)(4-morpholinyl)methanone
- (6R,7R)-7-Amino-3-[(1Z)-2-(4-methyl-5-thiazolyl)et
- (6R,7R)-7-Amino-3-(chloromethyl)-8-oxo-5-thia-1-az
- (6R,7R)-7-Amino-8-oxo-3-(1-propenyl)-5-thia-1-azab
- (6S)-5,6-Dihydro-6-methyl-4H-thieno[2,3-b]thiopyra
- (6S)-5-Azaspiro[2.4]heptane-5,6-dicarboxylic acid
- (6S)-6-[5-(7-Bromo-9,9-difluoro-9H-fluoren-2-yl)-1
- (6S)-6-[5-[7-[2-(1R,3S,4S)-2-Azabicyclo[2.2.1]hept
- (7a,17b)-7-(9-Bromononyl)-estra-1,3,5(10)-triene-3
- (7a,17b)-7-(9-Bromononyl)estra-1,3,5(10)-triene-3,
- (7a,17b)-7-[9-[(4,4,5,5,5-Pentafluoropentyl)thio]n
- (7alpha,17beta)-17-(Acetyloxy)-7-(9-bromononyl)est
- (7S,9S)9-Acetyl-7,8,9,10-Tetrahydro-6,7,9,11-Tetra
- (8a,9R)-Cinchonan-9-ol mono[[(S)-[(1R)-2-methyl-1-
- (9-[1,1'-Biphenyl]-4-yl-9H-carbazol-3-yl)boronic a
- (9,9-Diphenyl-9H-fluoren-2-yl)boronic acid
- (9b,11b,16a)-9,11-Epoxy-17,21-dihydroxy-16-methyl-
- (9b,11b,16a)-9,11-Epoxy-21-hydroxy-16-methylpregna
- (9-Phenyl-9H-carbazol-3-yl)boronic acid
- 3-[2-(Pyridin-2-yl)ethenyl]-1-(tetrahydro-2H-pyran
- (alphaR)-alpha-(2,4-Difluorophenyl)-alpha-[(1R)-1-
- (alphaR)-alpha-[[[2-(4-Aminophenyl)ethyl]amino]met
- (alphaR)-alpha-[[[2-(4-Nitrophenyl)ethyl]amino]met
- (alphaR)-alpha-Hydroxy-N-[2-(4-nitrophenyl)ethyl]b
- (alphaR)-alpha-[(Methoxycarbonyl)amino]benzeneacet
- (alphaR,gammaS)-gamma-[(3-Carboxy-1-oxopropyl)amin
- (alphaS)-3-Ethoxy-4-methoxy-alpha-[(methylsulfonyl
- (alphaS)-4-Amino-alpha-(3-Amino-3-Oxopropyl)-1,3-D
- (alphaS)-alpha-[[(1R)-2-Hydroxy-1-phenylethyl]amin
- (alphaS)-alpha-Amino-2-chloro-benzeneacetic acid m
- (alphaS)-alpha-Amino-3-ThiopheneAcetic Acid
- (alphaS)-alpha-Aminobenzenebutanoyl-L-leucyl-L-phe
- (alphaS)-alpha-Amino-N-[(1R)-1-[(3aS,4S,6S,7aR)-he
- (alphaS)-alpha-[[[Methyl[[2-(1-methylethyl)-4-thia
- (alphaS,betaR)-beta-(2,5-Difluorophenyl)-beta-hydr
- (alphaS,betaS)-beta-Amino-alpha-[[1-[[4-(2-pyridin
- (aR,3aS,4S,6S,7aR)-Hexahydro-3a,8,8-trimethyl-alph
- (Benzothiazol-2-yl)-(Z)-2-trityloxyimino-2-(2-amin
- (betaR)-beta-Cyclopentyl-4-(4,4,5,5-tetramethyl-1,
- (betaR)-beta-Cyclopentyl-4-[7-[[2-(trimethylsilyl)
- (betaR,gammaR)-gamma-Ethyl-3-methoxy-N,N,beta-trim
- (betaS)-beta-Amino-3-ethoxy-4-methoxybenzeneethano
- (Bromomethyl)cyclopropane
- (E)-1,4-Dibromobut-2-ene
- (E)-3-(3-(N-phenylsulfamoyl)phenyl)acrylic acid
- (E)-3-[2-Cyclopropyl-4-(4-fluorophenyl)-3-quinolin
- (E)-3-[3'-(4-Fluorophenyl)-1'-(1-methylethyl)-1H-i
- (E)-3-[3'-(4''-Fluorophenyl)-1'-(1''-methylethyl)-
- (E)-4-(4-Nitrophenyl)But-3-En-2-One
- (E)-6-Iodo-3-[2-(pyridin-2-yl)ethenyl]-1-(tetrahyd
- (E)-6-Nitro-3-[2-(pyridin-2-yl)ethenyl]-1-(tetrahy
- (E)-7-[2-Cyclopropyl-4-(4-fluorophenyl)-3-quinolin
- (E)-()-3-methyl-1-(2,6,6-trimethylcyclohex-1-en-1
- (E)-methyl6-(4-hydroxy-6-methoxy-7-methyl-3-oxo-1,
- (E)-N-(2-Chloro-6-methylphenyl)-3-ethoxyacrylamide
- (E)-triMethyl((1-iodo-4-Methyloct-1-en-4-yl)oxy)si
- (N,N-dimethyl-3-(naphthalen-1-yloxy)-3-(thiophen-2
- (Phenylazo)malonitrile
- (R) -3- amino-1- ( 3- ( trifluoromethyl ) -5, 6-di
- (R)-(-)-1,3-Butanediol
- (R)-(-)-1-Aminoindane hydrochloride
- (R)-(-)-2,2-Dimethyl-1,3-dioxolane-4-methanol
- (R)-(-)-2-Amino-1-butanol
- (R)-(-)-2-Amino-3-methyl-1-butanol
- (R)-(-)-2-Chloromandelic acid
- (R)-(-)-2-Hydroxymethylpyrrolidine
- (R)-(-)-3-Carbamoymethyl-5-methylhexanoic acid
- (R)-(-)-3-Chloro-1,2-propanediol
- (R)-(-)-3-Quinuclidinol
- (R)-(-)-4-Phenyl-2-oxazolidinone
- (R)-(-)-alpha-(Trifluoromethyl)benzylamine
- R)-(-)-alpha-Methoxyphenylacetic acid
- (R)-(-)-Epichlorohydrin
- (R)-(-)-gamma-Toluenesulfonylmethyl-gamma-Butyrolactone
- (R)-(-)-O-Formylmandeloyl chloride
- (R)-(+)-1-(1-Naphthyl)ethylamine
- (R)-(+)-1,1'-Bi-2-naphthol
- (R)-(+)-1-Benzyl-3-pyrrolidinol
- (R)-(+)-1-Boc-3-aminopyrrolidine
- (R)-(+)-1-Phenylethylamine
- (R)-(+)-2-(4-Hydroxyphenoxy)propionic acid
- (R)-(+)-2-(Diphenylhydroxymethyl)pyrrolidine
- (R)-(+)-2,2-Dimethyl-1,3-dioxolane-4-carboxaldehyde
- (R)-(+)-4-Isopropyl-2-oxazolidinone
- (R)-(+)-9-(2-Hydroxypropyl)adenine
- (R)-(+)-Glycidol
- (R)-(+)-N-Benzyl-1-phenylethylamine
- (R)-(+)-Propylene carbonate
- (R)-[3-(3-Fluoro-4-morpholinophenyl)-2-oxo-5-oxazolidinyl]methyl methanesulfonate
- (R)-1-(2,6-Dichloro-3-fluorophenyl)ethanol
- (R)-1,4-Dioxaspiro[4,5]decane-2-carboxaldehyde
- (R)-1-[(S)-2-(Diphenylphosphino)ferrocenyl]ethyldi-tert-butylphosphine
- (R)-1-[3,5-Bis(trifluoromethyl)phenyl]ethanol
- (R)-1-Boc-3-Aminopiperidine
- (R)-1-Boc-3-Hydroxypiperidine
- (R)-1-N-Boc-2-methylpiperazine
- (R)-2-[(5-Bromo-1H-indol-3-yl)carbonyl]-1-pyrrolidinecarboxylic acid benzyl ester
- (R)-2-[3-(Diisopropylamino)-1-phenylpropyl]-4-(hydroxymethyl)phenol fumaric acid salt
- (R)-2-Amino-N-benzyl-3-methoxypropanamide
- (R)-2-Hydroxybutyric acid
- (R)-2-Methyl-pyrrolidine
- (R)-2-Methylpyrrolidine hydrochloride
- (R)-3-(3-Methylbutanoyl)-4-benzyloxazolidin-2-one
- (R)-3-(4-Phenoxyphenyl)-1-(piperidin-3-yl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine
- (R)-3-(Boc-Amino)piperidine
- (R)-3-amino-1,2,3,4-tetrahydrocarbazole
- (R)-3-Amino-2-azepanone
- (R)-3-Amino-3-benzo[1,3]dioxol-5-ylpropionic acid
- (R)-3-Amino-4-(2,4,5-trifluorophenyl)butyric acid
- (R)-3-Aminopiperidine dihydrochloride
- (R)-3-Aminoquinuclidine dihydrochloride
- (R)-3-Hydroxypiperidine hydrocloride
- (R)-3-Quinuclidinol hydrochloride
- (R)-4-[4-[(3-Chloro-2-hydroxypropyl)amino]phenyl]morpholin-3-one
- (R)-4-Benzyl-2-oxazolidinone
- (R)-5-(Azidomethyl)-3-[3-fluoro-4-(4-morpholinyl)phenyl]-2-oxazolidinone
- (R)-5-Bromo-3-((1-methylpyrrolidin-2-yl)methyl)-1H-indole
- (R)-5-Hydroxymethyldihydrofuran-2-one
- (R)-6-(4-Aminophenyl)-4,5-dihydro-5-methyl-3(2H)-pyridazinone
- (R)-6-Fluoro-3,4-dihydro-2H-1-benzopyran-2-carboxylicacid
- (R)-alpha-Methoxy-alpha-(trifluoromethyl)phenylacetyl chloride
- (R)-alpha-Methyl-4-nitrobenzylamine
- (R)-Glycidyl butyrate
- (R)-Homo-beta-alanine hydrochloride
- (R)-Mandelic acid
- (R)-N-(2,3-Epoxypropan-1-yl)phthalimide
- (R)-N-Benzyl-2-(benzyloxycarbonylamino)-3-hydroxypropionamide
- (R)-N-Boc-(3-Pyridyl)alanine
- (R)-N-Boc-(4-Pyridyl)alanine
- (R)-O-Acetylmandelic acid chloride
- (R)-Styrene oxide
- (R)-Tetrahydropapaverine N-acetyl-L-leucinate
- (R,R)-(-)-N,N'-Bis(3,5-di-tert-butylsalicylidene)-1,2-cyclohexanediamine
- (R,R)-1,2-Bis(methanesulfonyloxymethyl)cyclohexane
- (R,R)-1,2-Cyclohexanedicarboxylic anhydride
- (R,R)-7-AMINO-3-(1-METHYLPYRROLIDINIO)METHYL-3-CEPHEM-4-CARBOXYLATE HCL
- (S)-((2-Guanidino-4-thiazolyl)methylisothiourea dihydrochloride
- (S)-(-)-1-(1-Naphthyl)ethylamine
- (S)-(-)-1-(4-Methoxyphenyl)ethylamine
- (S)-(-)-1,2,3,4-Tetrahydro-naphthoic acid
- (S)-(-)-1,2-Diaminopropane dihydrochloride
- (S)-(-)-1-Boc-2-pyrrolidinemethanol
- (S)-(-)-1-Boc-3-aminopyrrolidine
- (S)-(-)-2-(Diphenylhydroxymethyl)pyrrolidine
- (S)-(-)-2-Chloropropionic acid
- (S)-(-)-3-(Benzoylthio)-2-methylpropanoic acid
- (S)-(-)-3-Amino-3-phenylpropionic acid hydrochloride
- (S)-(-)-4-Amino-2-hydroxybutyric acid
- (S)-(-)-Indoline-2-carboxylic acid
- (S)-(-)-N,N-Dimethyl-3-hydroxy-3-(2-thienyl)propanamine
- (S)-(-)-N-Benzyl-1-phenylethylamine
- (S)-(+)-2,2-Dimethyl-1,3-dioxolane-4-methanol
- (S)-(+)-2,2-Dimethylcyclopropanecarboxamide
- (S)-(+)-2-Amino-3-methyl-1-butanol
- (S)-(+)-2-Chlorophenylglycine methyl ester
- (S)-(+)-2-Methylpiperazine
- (S)-(+)-2-Phenylglycinol
- Industrial Grade (S)-(+)-2-Phenylpropionic Acid
- (S)-(+)-3-Hydroxytetrahydrofuran
- (S)-(+)-3-Quinuclidinol
- (S)-(+)-Dihydro-5-(p-Tolylsulfonyloxymethyl)-2(3H)-Furanone
- (S)-(+)-Epichlorohydrin
- (S)-(+)-N-(2,3-Epoxypropyl)phthalimide
- (S)-(+)-O-Acetyl-L-mandelic acid
- (S)-[1-(3-Mercapto-1-pyrrolidinyl)ethylidene]carbamic acid (4-nitrophenyl)methyl ester
- (S)-1-(2,4-Dichlorophenyl)-1,2-ethanediol
- (S)-1-(2,4-Dichlorophenyl)-1,2-ethanediol dimethanesulfonate
- (S)-1-(2,6-Dichloro-3-fluorophenyl)ethanol
- (S)-1-(2-Amino-5-chlorophenyl)-1-(trifluoromethyl)-3-cyclopropyl-2-propyn-1-ol
- (S)-1-(2-Chloroacetyl)pyrrolidine-2-carboxamide
- (S)-1-(3-Ethoxy-4-methoxyphenyl)-2-(methylsulfonyl)ethylamine N-acetyl-L-leucine salt
- (S)-1-(3-Methoxyphenyl)ethylamine
- (S)-1-(4-Chlorophenyl)ethylamine
- (S)-1-(4-Nitrophenyl)ethylamine hydrochloride
- (S)-1,2,3,4-Tetrahydro-5-methoxy-N-propyl-2-naphthalenamine hydrochloride
- (S)-1-[(Acetylamino)methyl]-2-chloroethyl acetate
- (S)-10-Hydroxycamptothecin
- (S)-1-Amino-3-chloro-2-propanol hydrochloride
- (S)-1-Benzyl-3-pyrrolidinol
- (S)-1-Boc-2-cyanopyrrolidine
- (S)-1-Boc-3-hydroxypiperidine
- (S)-1-N-Boc-2-methylpiperazine
- (S)-2-(((9H-Fluoren-9-yl)methoxy)carbonylamino)-6-aminohexanoic acid
- (S)-2-(1,6,7,8-Tetrahydro-2H-indeno[5,4-b]furan-8-yl)ethylamine
- (S)-2-(3-((2-Isopropylthiazol-4-yl)methyl)-3-methylureido)-3-methylbutanoic acid
- (S)-2-(4-Fluorophenyl)-3-Methylbutyric Acid
- (S)-2-(Aminomethyl)-1-ethylpyrrolidine
- (S)-2-(Benzyloxycarbonylamino)-3-butenoic acid methyl ester
- (S)-2,2-Diphenyl-2-(pyrrolidin-3-yl)acetonitrile hydrobromide
- (S)-2,4-Dichloro-alpha-(chloromethyl)-benzenemethanol
- (S)-2-[(4-Chlorophenyl)(4-piperidinyloxy)methyl]pyridine
- (S)-2-Amino-2'-hydroxy-1,1'-binaphthyl
- (S)-2-Amino-5-methoxytetrahydronaphthalene hydrochloride
- (S)-2-Amino-5-methoxytetralin (S)-mandelate
- (S)-2-Amino-5-Methoxytetralin Hydrochloride
- (S)-2-Benzothiazolyl (Z)-2-(2-aminothiazole-4-yl)-2-methoxycarbonylmethoxyiminothioacetate
- (S)-2-Benzothiazolyl (Z)-2-(5-amino-1,2,4-thiadiazol-3-yl)-2-methoxyiminothioacetate
- (S)-2-Benzyl-N,N-dimethylaziridine-1-sulfonamide
- (S)-2-Benzylsuccinic acid
- (S)-2-chloro-1-(2,4-dichlorophenyl)ethyl methanesulfonate
- (S)-2-Chloromandelic acid
- (S)-2-Hydroxy-3-methoxy-3,3-diphenylpropionic acid
- (S)-2-Hydroxybutyric acid
- (S)-3-(1-Cyano-1,1-diphenylmethyl)-1-tosylpyrrolidine
- (S)-3-(2-Thienylthio)butanoic acid
- (S)-3-(4-Methoxy-3-sulfonamidophenyl)-2-propylamine Hydrochloride
- (S)-3-(Benzyloxycarbonyl)-4-isopropyl-2,5-oxazolidinedione
- (S)-3-(Boc-amino)-4-phenylbutyric acid
- (S)-3,4-Dihydro-6-chloro-4-hydroxy-2H-thieno[3,2-e]-1,2-thiazine-1,1-dioxide
- (S)-3-amino-1,2,3,4-terahydrocarbazole
- (S)-3-Amino-1-N-Boc-piperidine
- (S)-3-Amino-3-phenylpropan-1-ol
- (S)-3-Amino-3-phenylpropanoic acid ethyl ester hydrochloride
- (S)-3-Aminoquinuclidine dihydrochloride
- (S)-3-Dimethylamino-1-(3-methoxyphenyl)-2-methyl-1-propanone
- (S)-3-Hydroxypyrrolidine hydrochloride
- (S)-3-Methyl-1-(2-piperidinophenyl)butylamine N-acetylglutamate salt
- (S)-3-Phenoxybenzaldehyde cyanohydrin
- (S)-3-tert-Butylamino-1,2-propanediol
- (S)-4-(4-(5-(Aminomethyl)-2-oxooxazolidin-3-yl)phenyl)morpholin-3-one
- (S)-4-(4-Aminobenzyl)-2(1H)-oxazolidinone
- (S)-4-(4'-Nitrobenzyl)-1,3-oxazolidine-2-one
- (S)-4,5,6,7-Tetrahydro-2,6-benzothiazolediamine
- (S)-4-Benzyl-2-oxazolidinone
- (S)-4-Nitro-alpha-methylbenzylamine
- (S)-5-(Aminomethyl)-3-(3-fluoro-4-morpholinophenyl)oxazolidin-2-one
- (S)-5-(tert-Butoxycarbonyl)-5-azaspiro[2.4]heptane-6-carboxylic acid
- (S)-5,6,7,8-Tetrahydro-6-(propylamino)-1-naphthalenol hydrobromide
- (S)-5-[(Biphenyl-4-yl)methyl]-1-(2,2-dimethylpropionyl)pyrrolidin-2-one
- (S)-5-[(Biphenyl-4-yl)methyl]pyrrolidin-2-one
- (S)-5-Hydroxymethyldihydrofuran-2-one
- (S)-5-Methoxy-N-propyl-N-(2-(thiophen-2-yl)ethyl)-1,2,3,4-tetrahydronaphthalen-2-amine
- (S)-6-Fluoro-3,4-dihydro-2H-1-benzopyran-2-carboxylic acid
- (S)-6-Oxo-2-piperidinecarboxylic acid
- (S)-8-Chloro-1-methyl-2,3,4,5-tetrahydro-1H-3-benzazepine
- (S)-alpha,alpha-Diphenyl-3-pyrrolidineacetamide
- (S)-alpha-Ethyl-2-oxo-1-pyrrolidineacetic acid (R)-alpha-methylbenzenemethanamine salt
- (S)-N-[[3-[3-Fluoro-4-(4-morpholinyl)phenyl]-2-oxo-5-oxazolidinyl]methyl]phthalimide
- (S)-N-Boc-2-pyrrolidone-5-carboxylic acid tert-butyl ester
- (S)-N-Carbobenzyloxy-4-amino-2-hydroxybutyric acid
- (S)-Pyrrolidine-2-carbonitrile hydrochloride
- (S)-tert-Butyl 3-oxo-1-phenylpropylcarbamate
- (S,S)-2,8-Diazabicyclo[4.3.0]nonane
- (S,S)-2-Azabicyclo[3,3,0]-octane-3-carboxylic acid benzylester hydrochloride
- (S,S,S)-2-Azabicyclo[3,3,0]-octane-carboxylic acid benzylester hydrochloride
- (S,Z)-5-Amino-2-(dibenzylamino)-1,6-diphenylhex-4-en-3-one
- (SP-4-3)-Dimethyl(1,1,1-Trifluoro-2,4-Pentanedionato)-Gold
- (trans,trans)-4-broMo-4'-pentyl-1,1'-Bicyclohexane
- (trans,trans)-4'-Propyl-[1,1'-bicyclohexyl]-4-methanol
- (Z)-(2,4-Difluorophenyl)-4-piperidinylmethanone oxime
- (Z)-2-(2-Aminothiazol-4-yl)-2-(1-carboxy-1-methyl)ethoxyiminoacetic acid
- (Z)-2-(2-Aminothiazol-4-yl)-2-(tert-butoxycarbonylmethoxyimino)acetic acid
- (Z)-2-(2-Aminothiazol-4-yl)-2-carboxymethoxyiminoacetic acid
- (Z)-2-(2-tert-Butoxycarbonylaminothiazol-4-yl)-2-pentenoic acid
- (Z)-2-(2-Tritylaminothiazol-4-yl)-2-(2-tert-butoxycarbonylprop-2-oxyimino)acetic acid
- (Z)-2-Amino-alpha-[1-(tert-butoxycarbonyl)]-1-methylethoxyimino-4-thiazolacetic acid
- (Z)-2-Amino-alpha-propylidene-4-thiazoleacetic acid methyl ester hydrochloride
- (Z)-2-Methoxyimino-2-(furyl-2-yl) acetic acid ammonium salt
- (Z)-4-Chloro-2-[(2-methoxy-2-oxoethoxy) imino]-3-oxobutanoic acid
- (Z)-5-Amino-alpha-(ethoxyimino)-1,2,4-thiadiazole-3-acetic acid
- [(1R)-2-(Biphenyl-4-yl)-1-formylethyl]carbamic acid tert-butyl ester
- [(1S)-1-[[(2-Fluoro-6-nitrobenzoyl)phenylamino]carbonyl]propyl]carbamic acid 1,1-dimethylethyl ester
- [(1S)-3-Methyl-1-[[(2R)-2-methyloxiranyl]carbonyl]butyl]carbamic acid 1,1-dimethylethyl ester
- [(1S,2R)-2-[3,5-Bis(trifluoromethyl)phenyl]-2-hydroxy-1-methylethyl]carbamic acid phenylmethyl ester
- [(1S,3S,4S)-4-Amino-3-hydroxy-5-phenyl-1-(phenylmethyl)pentyl]carbamic acid 1,1-dimethylethyl ester
- [(2,6-Dimethylphenyl)Aminocarbonylmethyl]Chloride
- [(2-Methyl-1-propionylpropoxy)(4-phenylbutyl)phosphinoyl]acetic acid
- [(2S,5R)-5-(5-Methyl-2,4-Dioxopyrimidin-1-Yl)-2,5-Dihydrofuran-2-Yl]Methyl Benzoate
- [(3S,4R)-1-Ethyl-4-(4-Fluorophenyl)-3-Piperidinyl]Methanol
- [(7S)-5-(Phenylmethyl)-5-azaspiro[2.4]hept-7-yl]carbamic acid tert-butyl ester
- [[(Carboxymethyl)amino]methylene]propanedioic acid 1,3-diethyl ester
- [[2-(6-Amino-9H-purin-9-yl)ethoxy]methyl]phosphonic acid diethyl ester
- [2-Cyclopropyl-4-(4-fluorophenyl)-quinolin-3-ylmethyl]-triphenyl-phosphonium bromido
- [[4-(2-Methoxyethyl)phenoxy]methyl]oxirane
- [[4-[[2-(1-Methylethoxy)ethoxy]methyl]phenoxy]methyl]oxirane
- [[4-[2-(Cyclopropylmethoxy)ethyl]phenoxy]methyl]oxirane
- [[5-(3-Fluorophenyl)-2-pyridinyl]methyl]phosphonic acid diethyl ester
- [1,1'-Biphenyl]-3-carboxylic acid, 5'-chloro-2'-hydroxy-
- LT-BPK(BiMaprostLatanprost)
- [2-(2-Phenoxyethoxy)Ethyl]Diethylamine
- [2-(4-Aminophenyl)ethyl]carbamic acid tert-butyl ester
- [2,8-Bis(trifluoromethyl)-4-quinolinyl]-2-pyridinylmethanone
- [2-[(5-Chloro-benzooxazol-2-yl)(3-oxobutyl)amino]ethyl]carbamic acid tert-butyl ester
- 11beta,21-dihydroxypregna-1,4,16-triene-3,20-dione 21-acetate
- [2-[2-(triphenylmethyl)-2H-tetrazol-5-yl]phenyl]boronic acid
- 5-Methylcyclocytidine Hydrochlorine
- [2S,3S,5S]-2-Amino-3-hydroxy-5-tert-butyloxycarbonylamino-1,6-diphenylhexane succinate salt
- [3-(Dimethylamino)propyl]triphenylphosphonium bromide hydrobromide
- [4-(2-Phenyl-1H-benzimidazol-1-yl)phenyl]boronic acid
- [4-[4-(4-Hydroxyphenyl)-1-piperazinyl]phenyl]carbamic acid phenyl ester
- [5-Bromo-3-[(1R)-(2,6-dichloro-3-fluorophenyl)ethoxy]pyridin-2-yl]amine
- [6-Methyl-2-(4-methylphenyl)imidazo[1,2-a]pyridin-3-yl]acetonitrile
- [S-(R*,R*)]- 2,2,2-Trifluoro-N-(1-oxiranyl-2-phenylethyl)acetamide
- 1-({[(3S)-Tetrahydro-3-Furanyloxy]Carbonyl}Oxy)-2,5-Pyrrolidinedione
- 1-(1,4-Benzodioxane-2-carbonyl)piperazine
- 1-(2-(4-(Chloromethyl)phenoxy)ethyl)azepane hydrochloride
- 1-(2,2-Difluorobenzodioxol-5-yl)cyclopropanecarboxylic acid
- 1-(2,2-Dimethoxyethyl)-1,4-dihydro-3-methoxy-4-oxo-2,5-pyridinedicarboxylic acid 2-methyl ester
- 1-(2,3-Dichlorophenyl)piperazine
- 1-(2,3-Dichlorophenyl)piperazine Hydrochloride
- 1-(2,3-Dihydro-1,4-benzodioxin-2-yl)ethan-1-one
- 1-(2,3-Dihydro-1,4-benzodioxin-2-ylcarbonyl)piperazine hydrochloride
- 1-(2,5-Difluorophenyl)-2-(1H-1,2,4-triazol-1-yl)ethanone
- 1-(2,6-Dichlorophenyl)-2-indolinone
- 1-(2,6-Dichlorophenyl)indolin-2-one
- 1-(2-Amino-3-bromo-4-methoxyphenyl)ethanone
- 1-(2-Amino-3-Chlorophenyl)-Ethanone
- 1-(2-Amino-3-Methoxy-Phenyl)Ethanone
- 1-(2-Amino-3-Methylphenyl)-Ethanone
- 1-(2-Amino-4-bromo-5-fluorophenyl)ethanone
- 1-(2-Amino-4-chloro-5-fluorophenyl)ethanone
- 1-(2-AMino-4-nitro-phenyl)-ethanone
- 1-(2-Amino-5-bromo-4-chlorophenyl)ethanone
- 1-(2-Aminoethyl)pyrrolidine
- 1-(2-Benzyloxy-Ethyl)-Piperazine
- 1-(2-Bromoethoxy)-4-nitrobenzene
- 1-(2-Chloro-4-(4-chlorophenyl)butyl)-1H-imidazole
- 1-(2-Chloroethyl)-1H-1,2,4-triazole
- 1-(2-Chloroethyl)-2-Methoxybenzene
- 1-(2-Chloroethyl)-4-methylpiperazine dihydrochloride
- 1-(2-Chloroethyl)azepane hydrochloride
- 1-(2-Fluoro-4-nitrophenyl)piperazine
- 1-(2-Fluorobenzyl)-1H-pyrazolo[3,4-b]pyridine-3-carbonitrile
- 1-(2-Fluorobenzyl)-1H-pyrazolo[3,4-b]pyridine-3-carboxaMide
- 1-(2-Fluorobenzyl)-1H-pyrazolo[3,4-b]pyridine-3-carboximidamide hydrochloride
- 1(2H)-Phthalazinone
- 1-(2-Hydroxyethyl)-4-methylpiperazine
- 1-(2-Iodoethyl)-4-octylbenzene
- 1-(2-Methoxyethyl)piperazine
- 1-(2-Methoxyphenyl)piperazine Hydrobromide
- 1-(2-Methoxyphenyl)piperazine Hydrochloride
- 1-(2-Pyrimidyl)piperazine Hydrochloride
- 1-(3-Carboxypyrid-2-yl)-2-phenyl-4-methyl-piperazine
- 1-(3-Chlorophenyl)-4-(3-chloropropyl)piperazine
- 1-(3-Chlorophenyl)-4-(3-Chloropropyl)Piperazine HCL
- 1-(3-Chlorophenyl)-4-(3-chloropropyl)piperazine hydrochloride
- 1-(3-Chlorophenyl)piperazine Dihydrochloride
- 1-(3-Chlorophenyl)piperazine Hydrochloride
- 1-(3-Chlorophenyl)piperazinium Chloride
- 1-(3-Chloropropyl)-1,3-dihydro-2H-benzimidazol-2-one
- 1-(3-Chloropropyl)-4-methylpiperazine dihydrochloride
- 1-(3-Chloropropyl)piperidine Monohydrochloride
- 1-(3-Dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride
- 1-(3-ethoxy-4-methoxyphenyl)-2-(methylsulfonyl)ethanol
- 1-(3-Ethyl-4-hydroxymethyl-phenyl)ethanone
- 1-(3-Hydroxypropyl)-4-Methylpiperazine
- 1-(3-Methyl-1-phenyl-5-pyrazolyl)piperazine
- 1-(3-Pyridyl)-3-(dimethylamino)-2-propen-1-one
- 1-(4-(1-PHENETHYL-1H-IMIDAZOLE-2-CARBONYL)PIPERIDIN-1-YL)ETHANONE
- 1-(4,5-Dihydro-2-thiazolyl)-3-azetidinethiol hydrochloride
- 1-(4-Amino-2-methylbenzoyl)-7-chloro-1,2,3,4-tetrahydro-5H-1-benzazepin-5-one
- 1-(4-Amino-3-Bromo-Phenyl)-Ethanone
- 1-(4-Aminophenyl)-2,3-Dihydro-1,3,3-Trimethyl-1H-Inden-5-Amine
- 1-(4-Aminophenyl)-2-Bromoethanone
- 1-(4-Aminophenyl)-4-(4-methoxyphenyl)piperazine
- 1-(4-Aminophenyl)-5,6-dihydro-3-(4-morpholinyl)-2(1H)-pyridinone
- 1-(4-Bromo-3-fluorophenyl)ethanone
- 1-(4-Bromophenyl)-2-phenylbenzimidazole
- 1-(4-BROMO-PHENYL)-PIPERIDIN-2-ONE
- 1-(4-Chlorobenzhydryl)piperazine
- 1-(4-Chlorophenyl)-1-cyclobutanecarbonitrile
- 1-(4-Chlorophenyl)-2-cyclopropylpropan-1-one
- 1-(4-Chlorophenyl)-2H-pyrazolin-3-one
- 1-(4-Chlorophenyl)-4,4-dimethyl-3-pentanone
- 1-(4-Fluorobenzyl)-2-chlorobenzimidazole
- 1-(4-Fluorophenethyl)-4-piperidone
- 1-(4-Fluorophenyl)-1,3-dihydroisobenzofuran-5-carbonitile
- 1-(4-Fluorophenylcarbamoyl)cyclopropanecarboxylic acid
- 1-(4-Hydrazinophenyl)methyl-1,2,4-triazole
- 1-(4-Hydroxybutyl)-4-Methylpiperazine
- 1-(4-Iodophenyl)-2-piperidinone
- 1-(4-Iodophenyl)-3-morpholino-5,6-dihydropyridin-2(1H)-one
- 1-(4-Methoxyphenyl)-2-benzylaminopropane
- 1-(4-Methoxyphenyl)-2-phenylethanone
- 1-(4-Methoxyphenyl)-4-(4-nitrophenyl)piperazine
- 1-(4-Methoxyphenyl)piperazine Dihydrochloride
- 1-(4-Nitrophenyl)methyl-1,2,4-triazole
- 1-(4-Pyridyl)acetone
- 1-(5-Amino-2-fluorophenyl)ethanone
- 1-(5-Bromo-2-fluorophenyl)ethanone
- 1-(5-Bromo-2-methoxy-phenyl)adamantane
- 1-(6-Methylpyridin-3-yl)-2-[4-(methylsulfonyl)phenyl]ethanone
- 1-(7-Bromo-9,9-difluoro-9H-fluoren-2-yl)-2-chloroethanone
- 1-(Benzyloxy)-2-(Chloromethyl)Benzene
- 1-(Bromoacetyl)pyrene
- 1-(Cbz-amino)cyclopropanecarboxylic Acid
- 1-(Cyanomethyl)imidazole
- 1-(cyanomethyl)imidazole Hydrochloride
- 1-(Cyclopropylcarbonyl)piperazine
- 1-(Dimethylamino)-2-Methyl-3-Pentanone
- 1-(Dimethylamino)-3-[2-[2-(3-methoxyphenyl)ethyl]phenoxy]-2-propanol hydrochloride
- 1-(Diphenylmethyl)-3-hydroxyazetidine
- 1-(Diphenylmethyl)piperazine
- 1-(Ethoxycarbonyl)piperidine-4-carboxylic acid
- 1-(Hydroxycyclohexyl)-(4-methoxyphenyl)acetonitrile
- 1-(Hydroxymethyl)cyclopropaneacetonitrile
- 1-(Piperidin-4-yl)-1,3-dihydrobenzoimidazol-2-one
- 1-(tert-Butyl) 2-methyl (2R,4S)-4-hydroxypiperidine-1,2-dicarboxylate
- 1-(Toluene-4-sulfonyl)-2,5-dihydro-1H-pyrrole
- 1-(Trifluoromethyl)-1,2-benziodoxol-3(1H)-one
- 1,1'-(4-Chlorobutylidene)Bis(4-Fluorobenzene)
- 1,1'-(Azodicarbonyl)-dipiperidine
- 1,1,1-Trifluoro-2,3-epoxypropane
- 1,1,1-Trimethoxy-2-methylpropane
- 1,1,1-Trimethyl-N-(trimethylsilyl)silanamine
- 1,1,2,2-Tetrachloroethane
- 1,1,2-Trichloroethane
- 1,1,2-Trimethoxyethane
- 1,1,2-Trimethyl-1H-benz[e]indole
- 1,1,3,3-Tetramethoxypropane
- 1,10-Phenanthroline Hydrate
- 1,10-Phenanthroline-5,6-dione
- 1,1-Bis(4-diethylaminophenyl)-4,4-diphenyl-1,3-butadiene
- 1,1-Bis(hydroxymethyl)cyclopropane
- 1,1'-Carbonyldiimidazole
- 1,1-Cyclobutanedicarboxylic Acid
- 1,1-Cyclohexanediacetic Acid
- 1,1-Cyclohexanediacetic acid mono amide
- 1,1-Dimethoxy-2-(2-methoxyethoxy)ethane
- 1,1-Dimethyl-1,2-ethanediamine
- 1,1-Dimethylpropargylamine
- 1,1-Diphenylethanol
- 1,1'-Thiocarbonyldiimidazole
- 1,2,3,4a,9,9a-Hexahydro-9-methyl-4H-carbazole-4-one
- 1,2,3,4-Tetrahydro-1-naphthoic Acid
- 1-(4-chloro-3-(trifluoromethyl)phenyl)-3-hydroxyurea
- CAUSTIC SODA LYE
- CAUSTIC SODA FLAKE
- Industrial Grade POTASSIUM PERMANGANATE
- Industrial Grade BORIC ACID
- HP- Phosphinidene
- KOH - Potassium hydroxide
- Chloroform .
- MDC - Dichloromethane
- POCl3 .
- TC -Technetium
- Industrial Chemicals
- Chemicals
- Fine chemicals
- Speciality chemicals
- Nutraceuticals & Food chemicals
- Dyes chemicals
- Citric Acid
- Octanol
- Herbal extracts & chemicals
- Para Octyl Phenol
- Potassium Phthalimide
- Pyrrolidine
- Pyridinium P Toluenesulfonate
- Sodium Thiosulphate
- Potassium Hydroxide Pellets
- 2,4-dichlorophenol
- 12 hydroxy stearic acid
- Barium Sulfate
- Acetic Acid Chemical
- Acetone
- Acrylic acid
- Aluminum Chloride
- Ammonium Molybdate
- Basic chromium sulfate
- Boric acid
- Butyl carbitol
- Calcium Chloride Dihydrate
- Phosphorus Oxychloride
- Industrial Grade Phosphorus Pentachloride
- Phosphorus Pentasulfide
- phosphorus pentoxide
- Phosphorous Tri Chloride
- Potassium Carbonate
- Potassium Carbonate Granules
- Potassium Iodide
- Potassium Permanganate
- Propylene Glycol
- Pyridine
- resolving agents
- Soap noodles
- Sodium Bisulfite
- Sodium Gluconate
- Sodium Hydroxide Pellets
- Sodium Iodide
- Sodium Metabisulphite
- Sodium Methoxide Solution
- Sodium methoxide powder solution
- Sodium Molybdate
- Sodium Stannate
- Sodium Sulphate
- Sodium Tungstate
- stannic chloride anhydrous
- Stannous Chloride Anhydrous
- Stannous Chloride Dihydrate
- Iodine
- Thionyl chloride
- Butylated hydroxytoluene (bht)
- Iso Butanol
- Calcium Chloride
- Caustic Potash Flakes
- Isophorone
- Chloroform
- Chlorosulphonic Acid
- Cobalt Acetate
- Isopropyl alcohol
- Cobalt chloride
- Bromine Solution
- Cobalt Sulfate
- Copper Sulfate
- Cyclohexane
- liquid cyloride
- cyclohexanone
- Industrial Grade Dicyandiamide
- Liquid Toluene
- dimethyl formamide
- Dimethyl sulfoxide
- lithium carbonade
- Dimethyl Sulphate
- Lithium Chloride
- Lithium Molybdate
- Lithium Nitrate
- Litium bromine
- Malinonitrile .
- ethyl acetate
- Formaldehyde
- Methylene Chloride
- Methyl Ethyl Ketone
- Methyl Isobutyl Ketcone
- Methyl Salicylate
- Nickel Metal
- Nickel sulpate
- N- methylpiperazine
- Nonylphenol
- normal butanol
- Formic Acid
- Industrial Grade Octanol
- Hydrogen Peroxide
- Oleum
- Ortho chloro penol
- Hydroxylamine Hydrochloride
- Oxalic Acid
- Oxalyl Cloride
- Tetrahydrofuran
- Paraffin Wax
- Tetrasodium Pyrophosphate
- Phosphoric Acid
- phosphorous oxychloride
- Phosphorus pentachloride
- phosphorus pentasulphide
- Trimethyl Orthoformate
- trimethyl sulfoxonium iodide
- Zinc Chloride Powder
- Zinc Oxide
- Stannous Fluoride
- Stannous Oxalate
- Stannous Oxide
- Stannous Sulphate
- Stearic Acid
- Sulfur Powder
- Sulfuryl Chloride
- Sulphanilic acid
- Sulphur Dioxide
- Sulphur Granules
- Sulphuric Acid
- Sulphuryl Chloride
- Asparaginase
- Barnidipine hydrochloride
- Berberine hydrogen sulphate
- Bexarotene
- Buparvaquone
- Cefozopran Hydrochloride
- Dextropropoxyphene Hydrochloride
- 1,1,1-Trichloro-2-methyl-2-propanol Hemihydrate
- Intermediates
- Pharmaceutical Excipients
- Herbal Extracts
- Nutraceutical Ingredients
- Active Pharmaceutical Ingredient
- Industry Served
- Contact Us
(6R,7R)-7-Amino-8-oxo-3-(1-propenyl)-5-thia-1-azab
Product Details:
- Poisonous Non-Poisonous
- Physical Form Solid
- Application Used as a key intermediate in cephalosporin antibiotic synthesis
- Shelf Life 24 months
- Appearance White to off-white crystalline powder
- Density 1.42 Gram per cubic centimeter(g/cm3)
- Taste Neutral
- Click to View more
X
(6R,7R)-7-Amino-8-oxo-3-(1-propenyl)-5-thia-1-azab Price And Quantity
- 25g, 100g, 500g, 1kg, or per request
- +120° to +130° (c=1, MeOH)
- Not classified as hazardous
- Stable under recommended conditions
- In accordance with pharmacopeia requirements
- Not regulated under ADR/RID
- Enantioselective synthesis
(6R,7R)-7-Amino-8-oxo-3-(1-propenyl)-5-thia-1-azab Product Specifications
- Pharmaceutical Intermediate
- >98%
- Odorless
- Crystalline
- Chiral beta-amino acid core, essential intermediate for cephalosporin antibiotics
- 228.27 g/mol
- Neutral
- (6R,7R)-7-Amino-8-oxo-3-(1-propenyl)-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid
- Pharmaceutical Grade
- Used as a key intermediate in cephalosporin antibiotic synthesis
- Solid
- Non-Poisonous
- 1.42 Gram per cubic centimeter(g/cm3)
- White to off-white crystalline powder
- 24 months
- Pharmaceutical manufacturing, active pharmaceutical ingredient (API) synthesis
- C9H12N2O2S
- See molecular structure in image
- Soluble in DMSO, slightly soluble in water
- 29419090
- Store in cool, dry place, tightly sealed container
- 160-165C
- 87621-39-0
- Not determined
- 25g, 100g, 500g, 1kg, or per request
- +120° to +130° (c=1, MeOH)
- Not classified as hazardous
- Stable under recommended conditions
- In accordance with pharmacopeia requirements
- Not regulated under ADR/RID
- Enantioselective synthesis
Product Description
CAS number: 120709-09-3
Properties :
| Synonym | 7-APRA |
| Molecular Formula | C10H12N2O3S |
| Molecular Weight | 240.28 |
| Specifications | AVAILABLE ON REQUEST |
| Packing | EXPORT WORTHY PACKING |
| Availability | FOR R & D PURPOSE ONLY |
Pharmaceutical-Grade Intermediate
This compound serves as an essential building block in the creation of cephalosporin antibiotics. Its high purity and precise optical rotation ensure batch-to-batch consistency, making it ideal for pharmaceutical manufacturing and active pharmaceutical ingredient synthesis. The products robust stability under recommended storage conditions supports a reliable two-year shelf life.
Safety and Quality Assurance
Manufactured through an enantioselective process, the product adheres to pharmacopeia requirements and features no hazardous classifications. Its non-poisonous nature and absence of strong odor make it safe for general handling and storage. Packing options accommodate different scales of operation, and each batch is quality-assured for pharmaceutical applications.
FAQs of (6R,7R)-7-Amino-8-oxo-3-(1-propenyl)-5-thia-1-azab:
Q: How should (6R,7R)-7-Amino-8-oxo-3-(1-propenyl)-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid be stored for optimal stability?
A: Store the compound in a cool, dry place, tightly sealed in its original container. Under these recommended conditions, the product remains stable and maintains its quality for up to 24 months.Q: What is the primary use of this chiral intermediate?
A: This product is primarily utilized as a key intermediate in the synthesis of cephalosporin antibiotics, making it essential in pharmaceutical manufacturing and active pharmaceutical ingredient (API) preparation.Q: When is this compound considered beneficial in pharmaceutical processes?
A: It is especially valuable during the synthesis of beta-lactam antibiotics, where its high chemical purity and optical activity enable the preparation of advanced cephalosporin derivatives.Q: Where can this compound be supplied from, and who are the end users?
A: The compound is exported, manufactured, and supplied from India. Typical end users include pharmaceutical companies, research laboratories, and chemical synthesis units involved in antibiotic development.Q: What is the manufacturing process used for this compound?
A: Enantioselective synthesis is employed, ensuring the correct chiral configuration, high purity, and pharmaceutical-grade quality suitable for incorporation into key cephalosporin structures.Q: How is this compound typically transported?
A: Transport of this compound is not regulated under ADR/RID, simplifying logistics and enabling safe and efficient worldwide distribution under ambient conditions.Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email




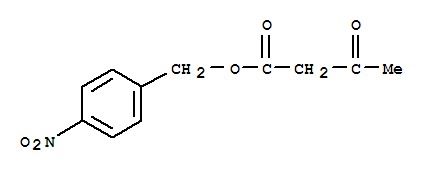
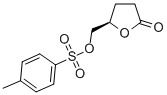


 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese